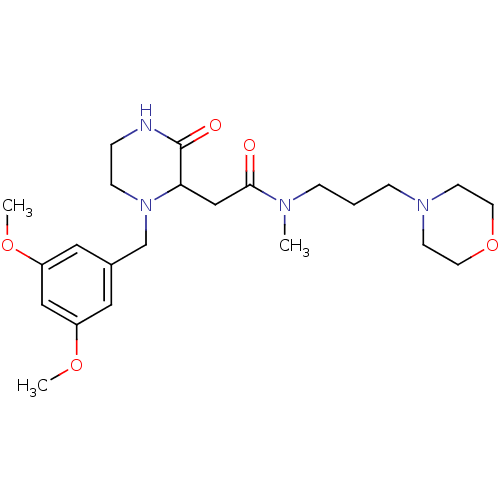
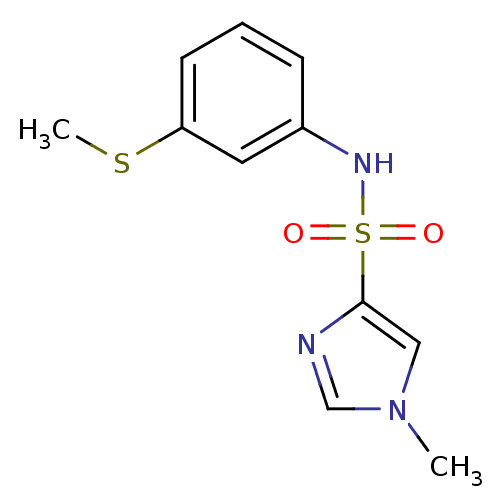
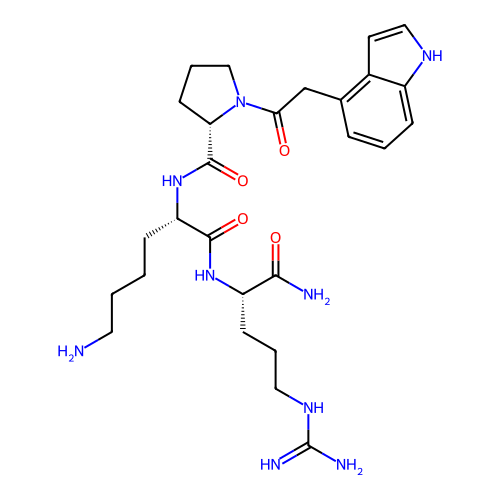
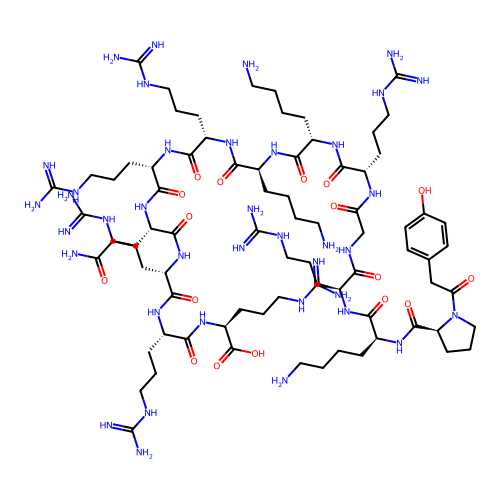
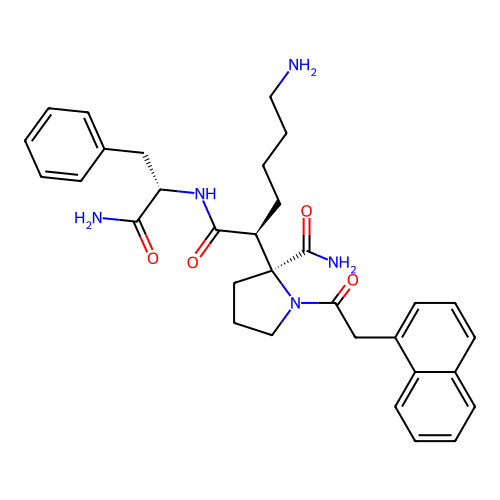
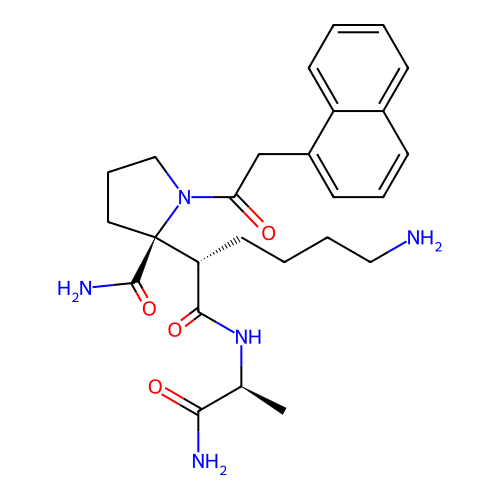
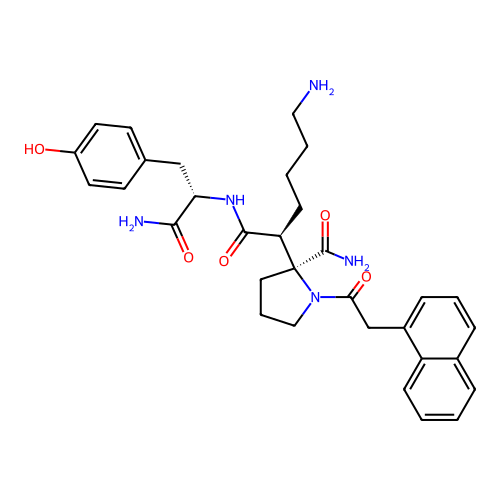
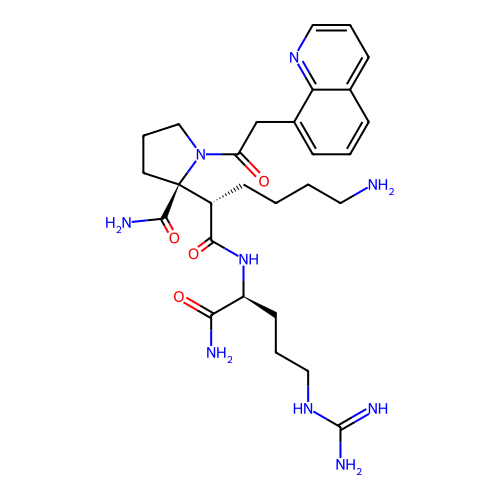
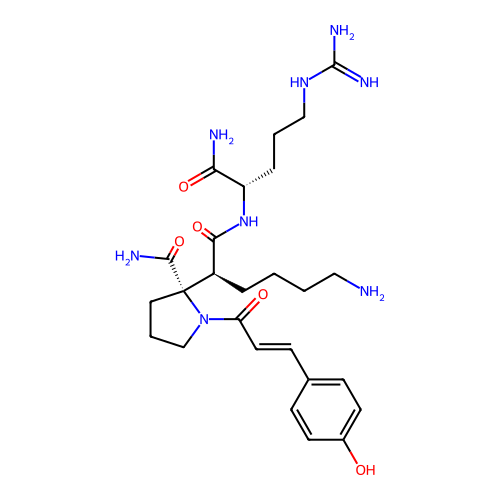
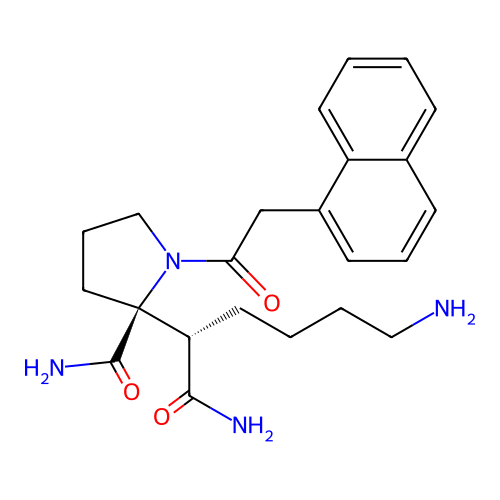
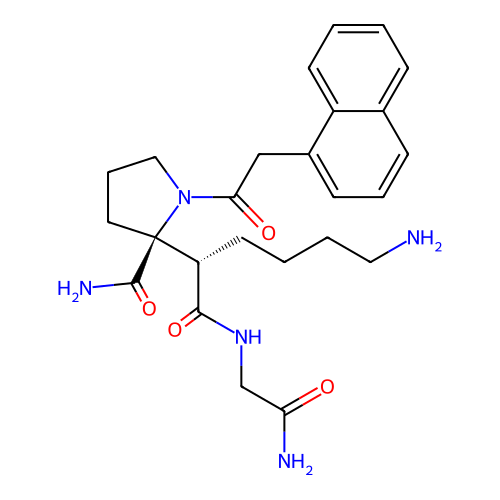
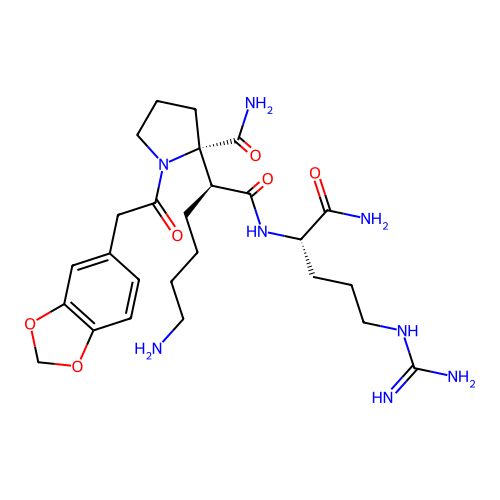
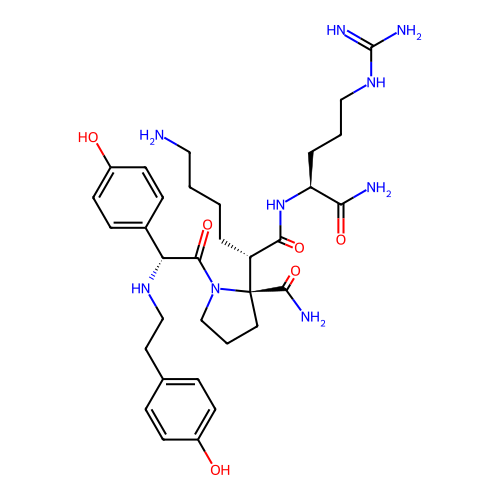
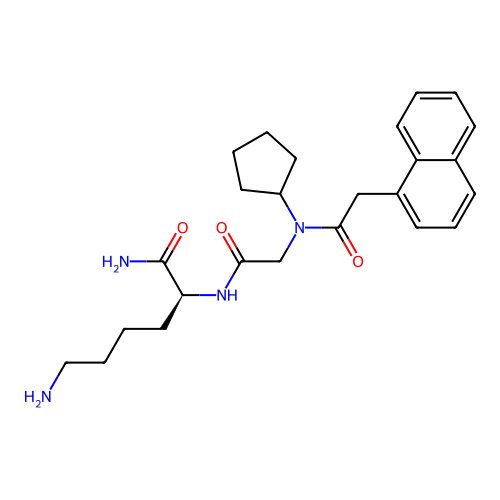
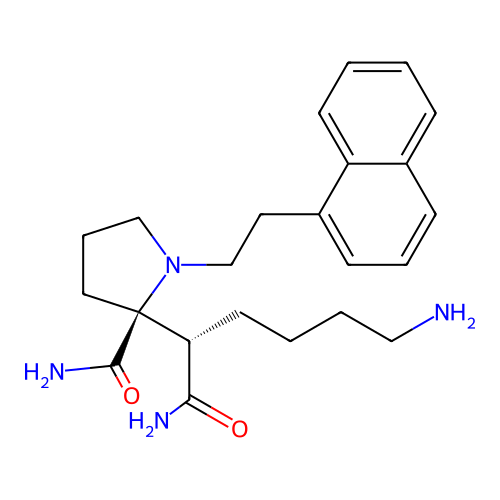
Report error Found 48 Enz. Inhib. hit(s) with all data for entry = 2838
Affinity DataIC50: 40nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 54nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 60nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 65nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 68nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 79nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 86nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 90nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 95nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 110nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 160nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 230nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 320nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 400nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 590nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 670nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 840nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 960nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 960nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 1.20E+3nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 1.30E+3nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 1.40E+3nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 1.50E+3nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 1.56E+3nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 1.60E+3nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 1.62E+3nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 1.63E+3nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 1.77E+3nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 2.50E+3nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 2.80E+3nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 3.13E+3nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+3nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 5.76E+3nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 8.96E+3nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 1.60E+4nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 2.07E+4nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 3.30E+4nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+5nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+5nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+5nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+5nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+5nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+5nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+5nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+5nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+5nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair