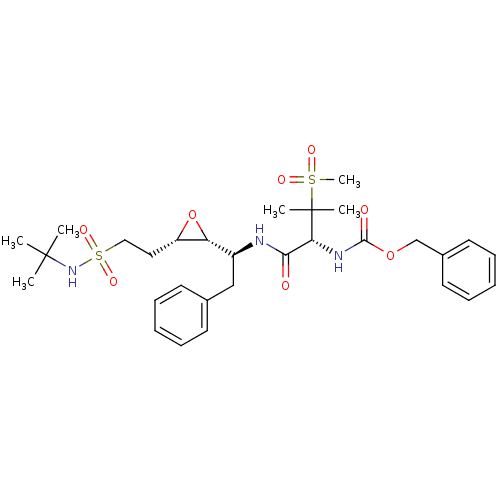
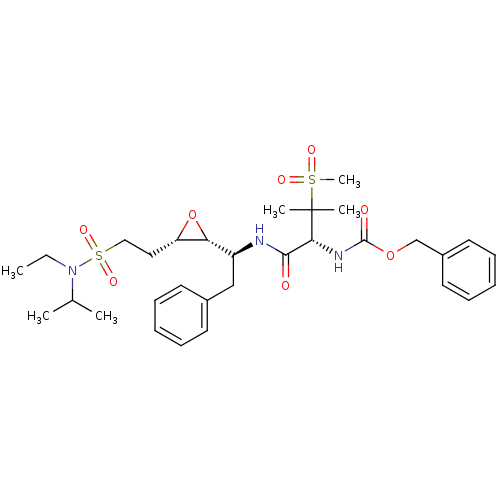
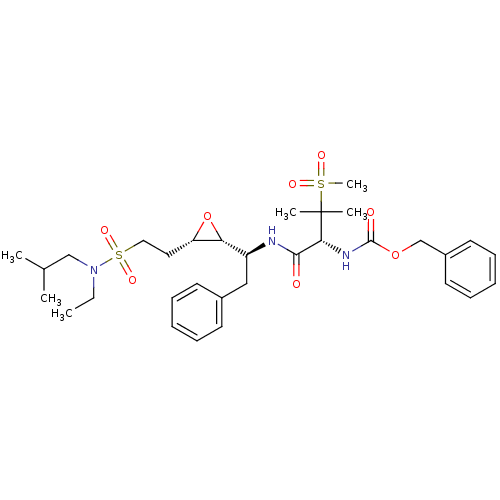
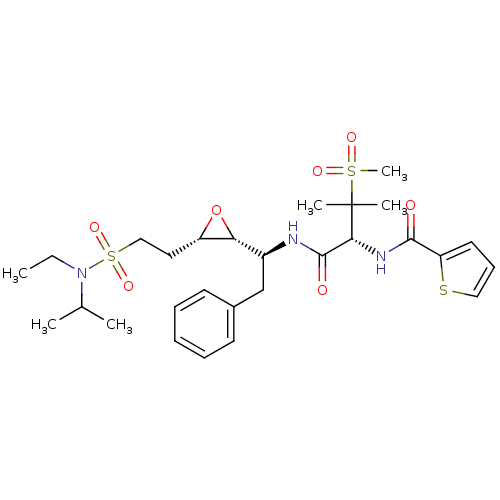
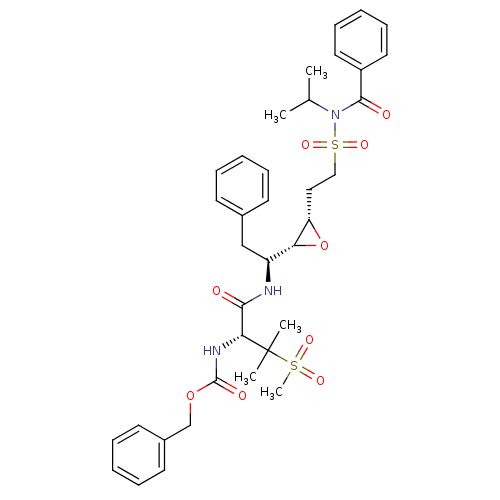
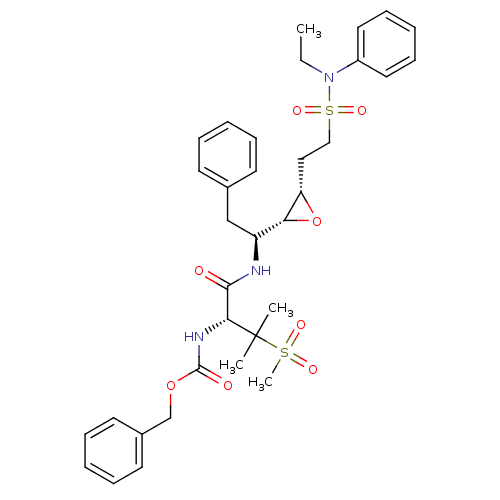
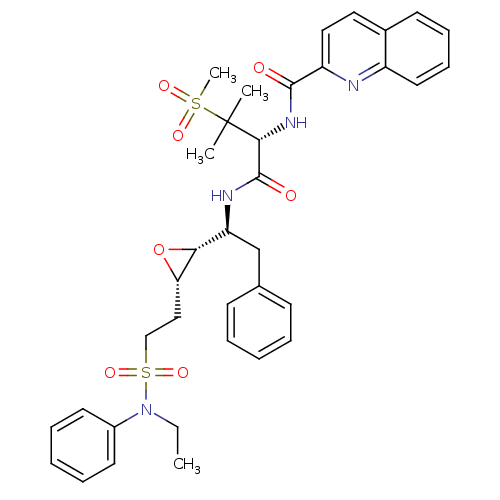
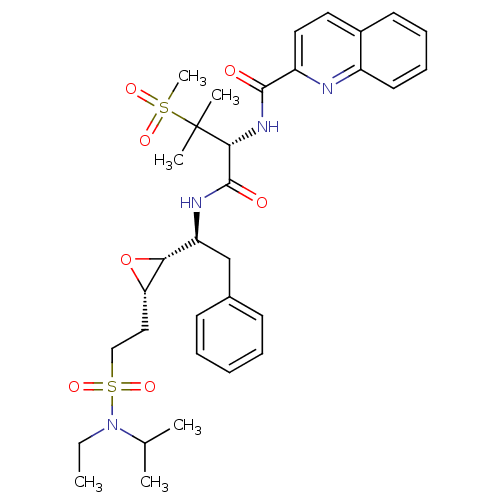
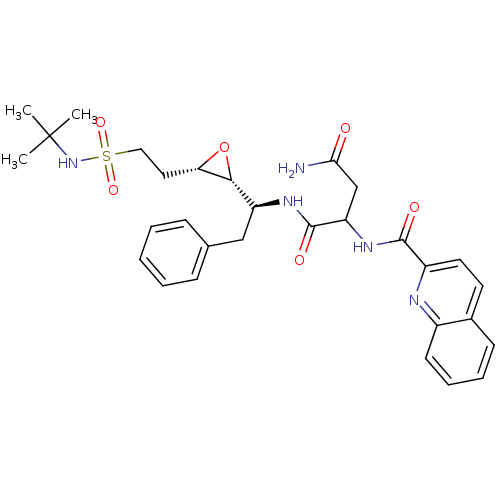
Report error Found 10 Enz. Inhib. hit(s) with all data for entry = 50030040
Affinity DataKi: 0.700nMAssay Description:Compound was tested for its inhibitory activity against HIV-1 proteaseMore data for this Ligand-Target Pair
Affinity DataKi: 0.800nMAssay Description:Compound was tested for its inhibitory activity against HIV-1 proteaseMore data for this Ligand-Target Pair
Affinity DataKi: 2.90nMAssay Description:Compound was tested for its inhibitory activity against HIV-1 proteaseMore data for this Ligand-Target Pair
Affinity DataKi: 3.10nMAssay Description:Compound was tested for its inhibitory activity against HIV-1 proteaseMore data for this Ligand-Target Pair
Affinity DataKi: 3.60nMAssay Description:Compound was tested for its inhibitory activity against HIV-1 proteaseMore data for this Ligand-Target Pair
Affinity DataKi: 4.60nMAssay Description:Compound was tested for its inhibitory activity against HIV-1 proteaseMore data for this Ligand-Target Pair
Affinity DataKi: 5nMAssay Description:Compound was tested for its inhibitory activity against HIV-1 proteaseMore data for this Ligand-Target Pair
Affinity DataKi: 7nMAssay Description:Compound was tested for its inhibitory activity against HIV-1 proteaseMore data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:Compound was tested for its inhibitory activity against HIV-1 proteaseMore data for this Ligand-Target Pair
Affinity DataIC50: 8.20E+3nMAssay Description:Inhibition against human cathepsin D was determinedMore data for this Ligand-Target Pair