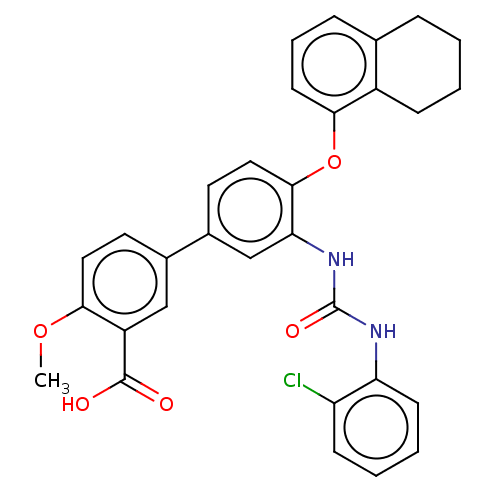
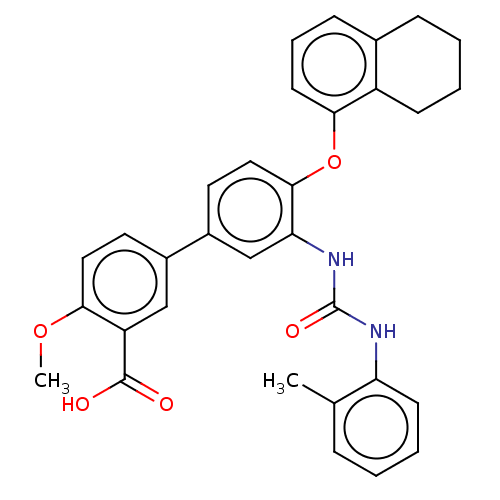
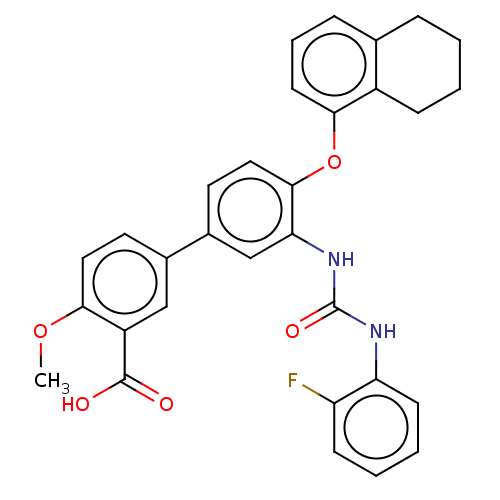
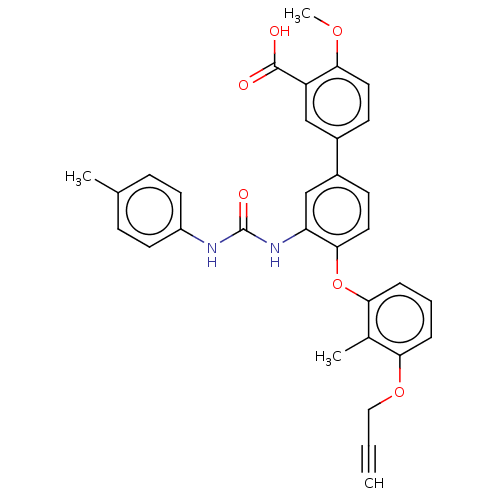
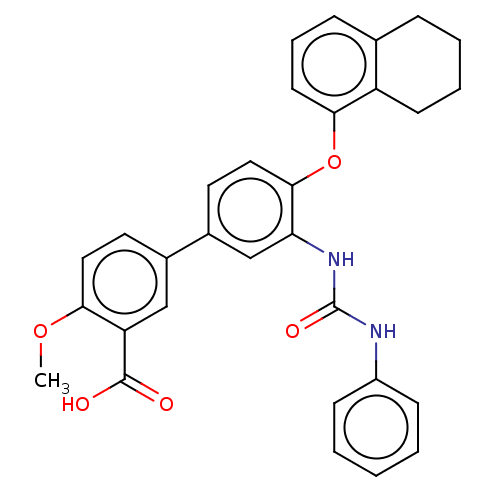
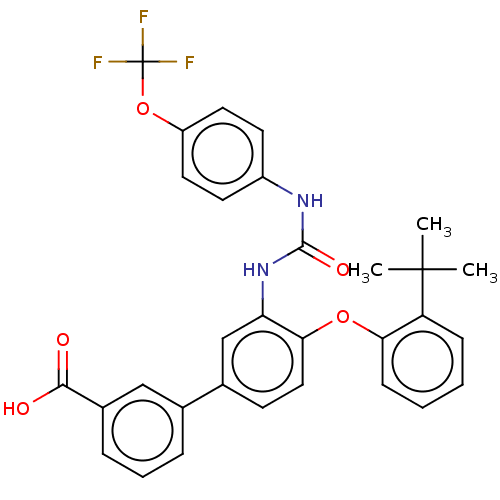
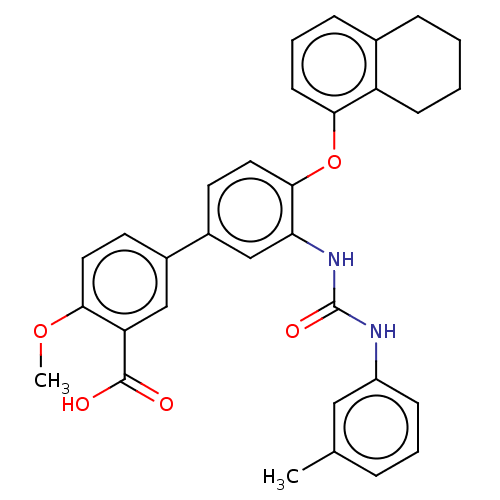
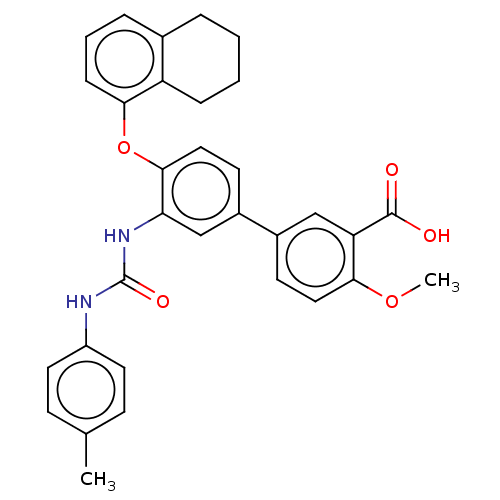
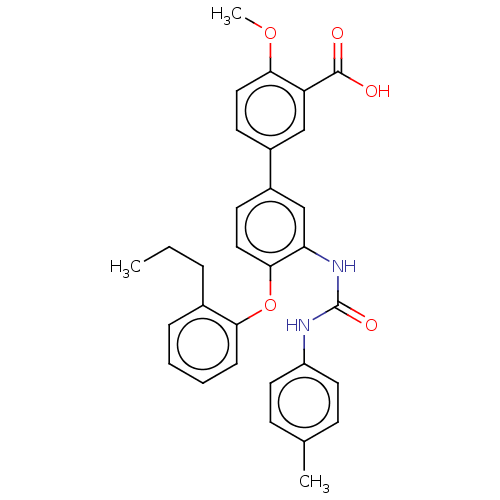
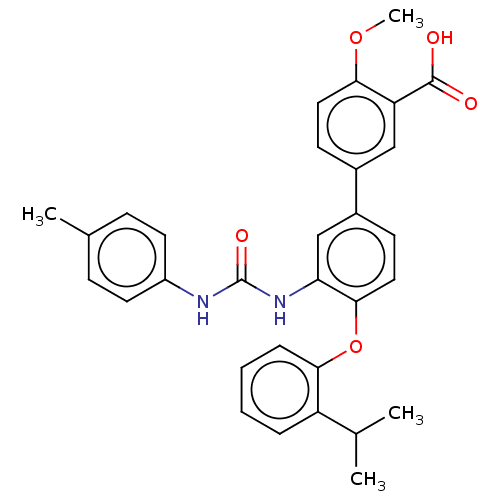
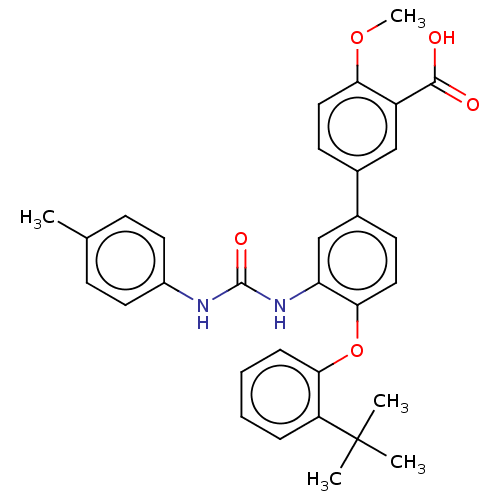
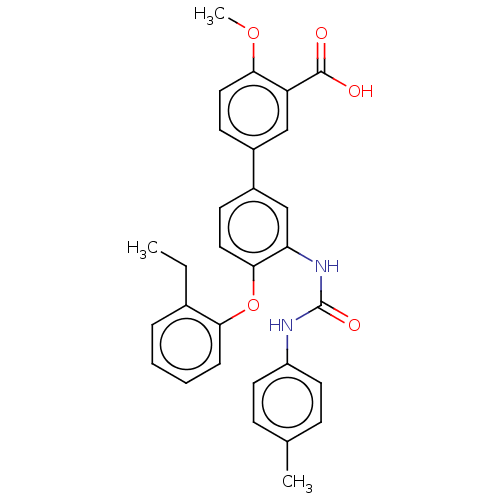
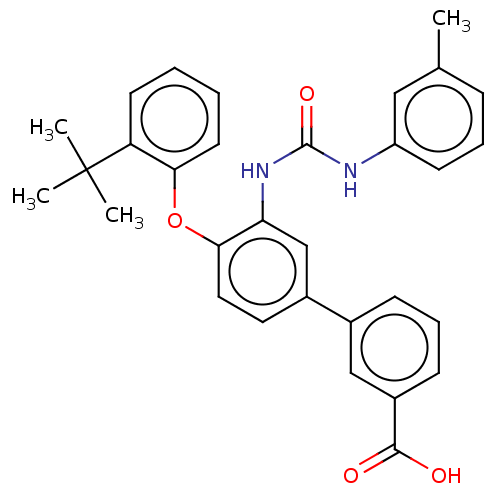
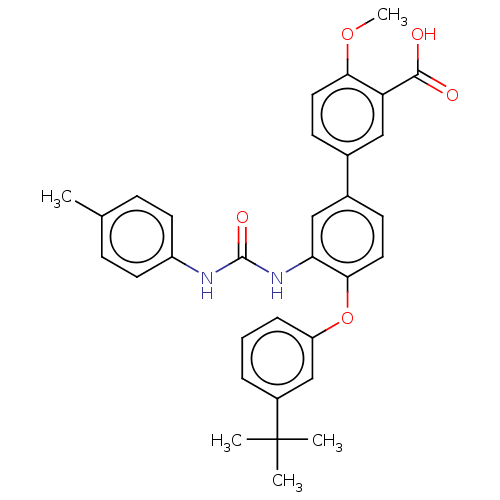
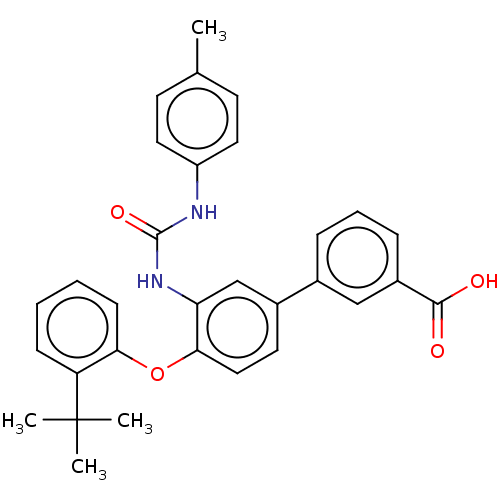
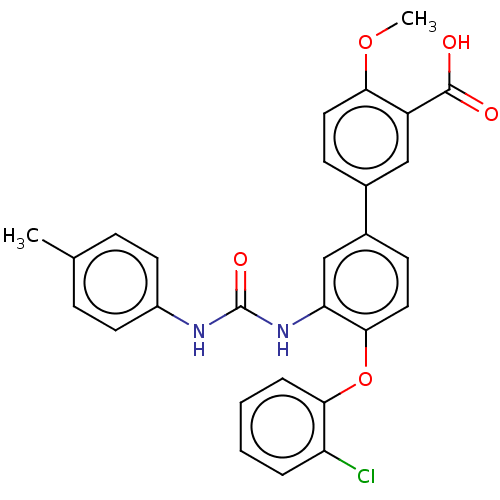
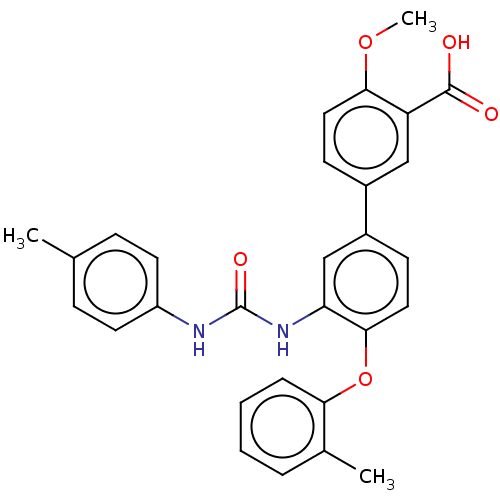
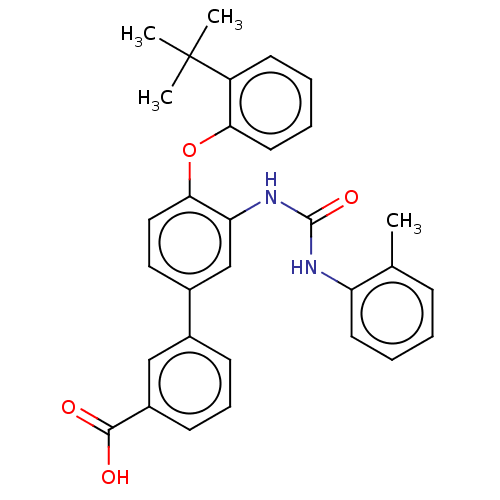
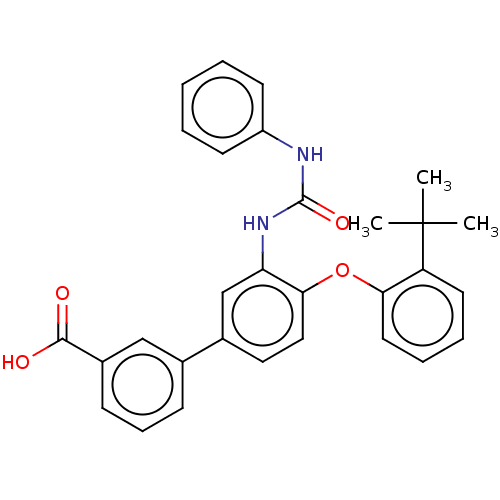
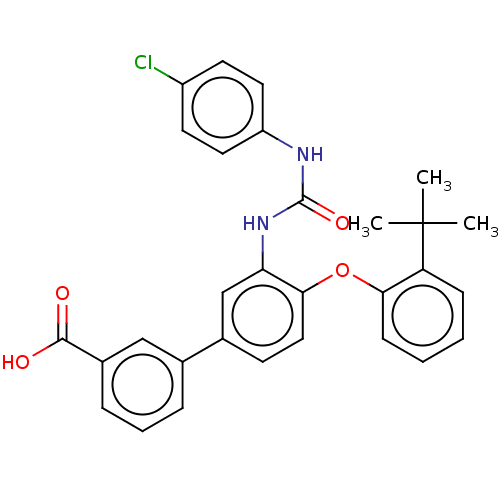
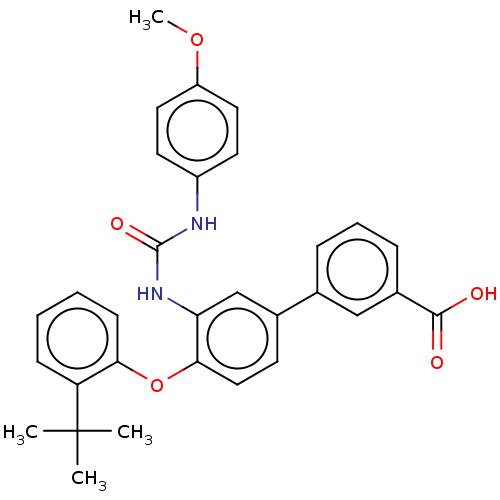
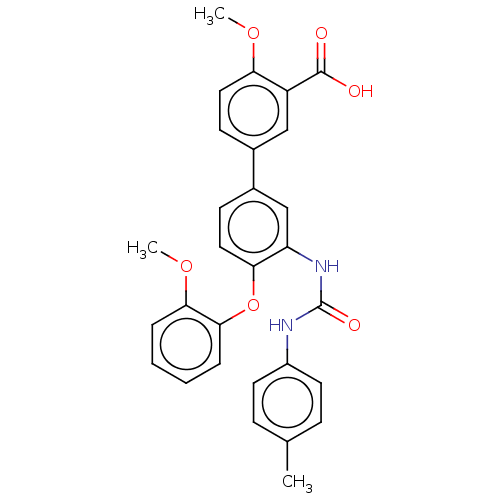
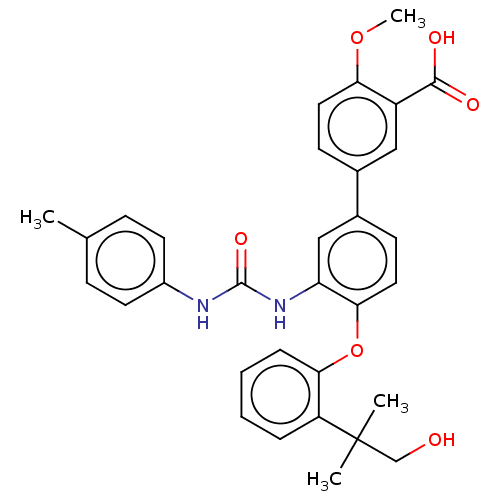
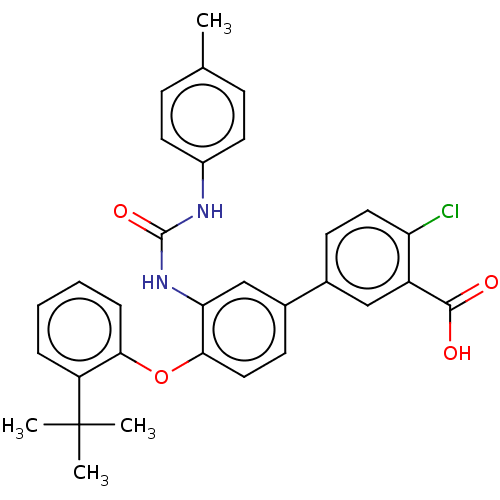
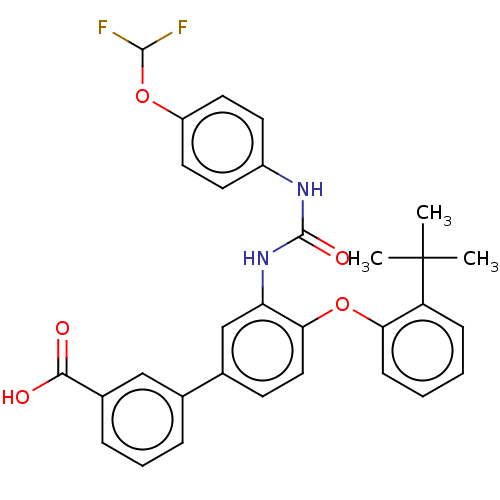
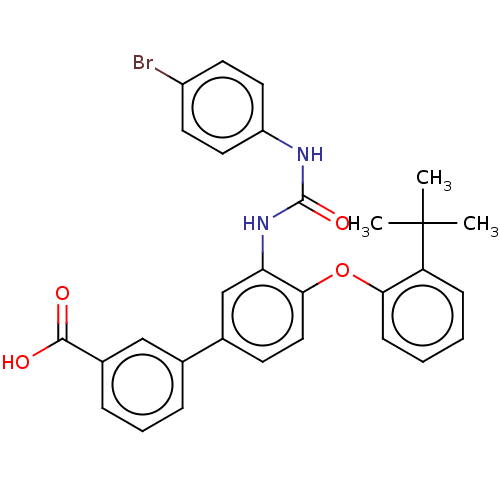
Report error Found 65 Enz. Inhib. hit(s) with all data for entry = 50049075
TargetIndoleamine 2,3-dioxygenase 1(Human)
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
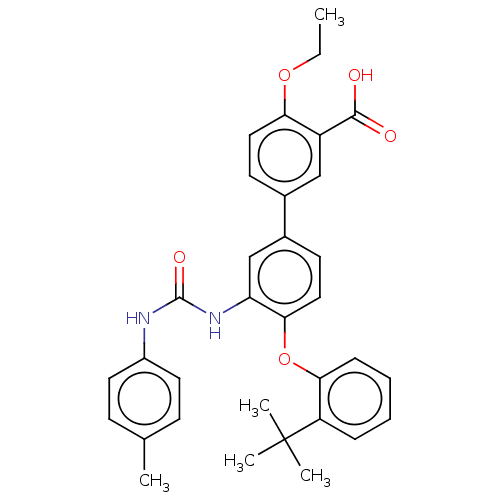
Affinity DataIC50: 22nMAssay Description:Inhibition of IDO1 in IFN-gamma stimulated human HeLa cells assessed as inhibition of kynurenine production preincubated with cells followed by IFN-g...More data for this Ligand-Target Pair
TargetIndoleamine 2,3-dioxygenase 1(Human)
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
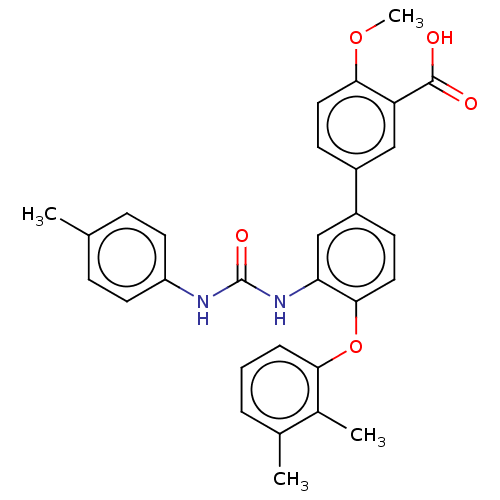
Affinity DataIC50: 49nMAssay Description:Inhibition of IDO1 in IFN-gamma stimulated human HeLa cells assessed as inhibition of kynurenine production preincubated with cells followed by IFN-g...More data for this Ligand-Target Pair
TargetIndoleamine 2,3-dioxygenase 1(Human)
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
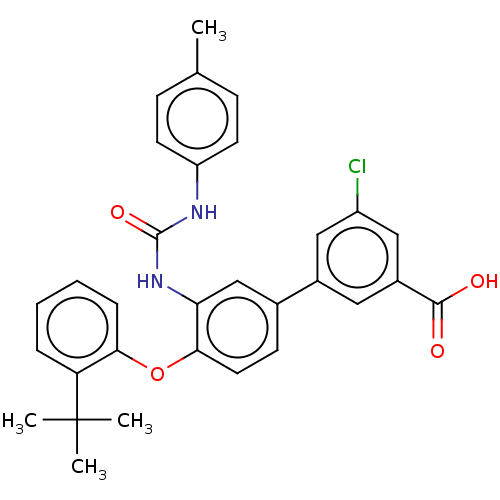
Affinity DataIC50: 50nMAssay Description:Inhibition of IDO1 in IFN-gamma stimulated human HeLa cells assessed as inhibition of kynurenine production preincubated with cells followed by IFN-g...More data for this Ligand-Target Pair
TargetIndoleamine 2,3-dioxygenase 1(Human)
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
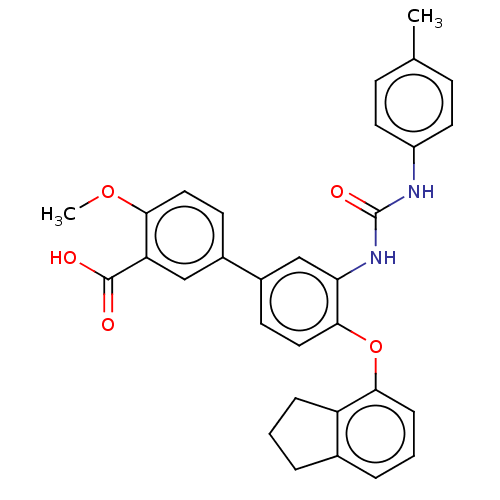
Affinity DataIC50: 50nMAssay Description:Inhibition of IDO1 in IFN-gamma stimulated human HeLa cells assessed as inhibition of kynurenine production preincubated with cells followed by IFN-g...More data for this Ligand-Target Pair
TargetIndoleamine 2,3-dioxygenase 1(Human)
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Affinity DataIC50: 62nMAssay Description:Inhibition of IDO1 in IFN-gamma stimulated human HeLa cells assessed as inhibition of kynurenine production preincubated with cells followed by IFN-g...More data for this Ligand-Target Pair
TargetIndoleamine 2,3-dioxygenase 1(Human)
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Affinity DataIC50: 87nMAssay Description:Inhibition of IDO1 (unknown origin) using tryptophan as substrate preincubated for 15 mins followed by substrate addition by high-throughput screenin...More data for this Ligand-Target Pair
TargetIndoleamine 2,3-dioxygenase 1(Human)
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Affinity DataIC50: 100nMAssay Description:Inhibition of IDO1 in IFN-gamma stimulated human HeLa cells assessed as inhibition of kynurenine production preincubated with cells followed by IFN-g...More data for this Ligand-Target Pair
TargetIndoleamine 2,3-dioxygenase 1(Human)
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Affinity DataIC50: 100nMAssay Description:Inhibition of IDO1 in IFN-gamma stimulated human HeLa cells assessed as inhibition of kynurenine production preincubated with cells followed by IFN-g...More data for this Ligand-Target Pair
TargetIndoleamine 2,3-dioxygenase 1(Human)
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Affinity DataIC50: 120nMAssay Description:Inhibition of IDO1 in IFN-gamma stimulated human HeLa cells assessed as inhibition of kynurenine production preincubated with cells followed by IFN-g...More data for this Ligand-Target Pair
TargetIndoleamine 2,3-dioxygenase 1(Human)
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Affinity DataIC50: 160nMAssay Description:Inhibition of IDO1 in IFN-gamma stimulated human HeLa cells assessed as inhibition of kynurenine production preincubated with cells followed by IFN-g...More data for this Ligand-Target Pair
TargetIndoleamine 2,3-dioxygenase 1(Human)
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Affinity DataIC50: 250nMAssay Description:Inhibition of IDO1 in IFN-gamma stimulated human HeLa cells assessed as inhibition of kynurenine production preincubated with cells followed by IFN-g...More data for this Ligand-Target Pair
TargetIndoleamine 2,3-dioxygenase 1(Human)
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Affinity DataIC50: 280nMAssay Description:Inhibition of IDO1 in IFN-gamma stimulated human HeLa cells assessed as inhibition of kynurenine production preincubated with cells followed by IFN-g...More data for this Ligand-Target Pair
TargetIndoleamine 2,3-dioxygenase 1(Human)
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Affinity DataIC50: 330nMAssay Description:Inhibition of IDO1 in IFN-gamma stimulated human HeLa cells assessed as inhibition of kynurenine production preincubated with cells followed by IFN-g...More data for this Ligand-Target Pair
TargetIndoleamine 2,3-dioxygenase 1(Human)
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Affinity DataIC50: 340nMAssay Description:Inhibition of IDO1 in IFN-gamma stimulated human HeLa cells assessed as inhibition of kynurenine production preincubated with cells followed by IFN-g...More data for this Ligand-Target Pair
TargetIndoleamine 2,3-dioxygenase 1(Human)
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Affinity DataIC50: 350nMAssay Description:Inhibition of IDO1 in IFN-gamma stimulated human HeLa cells assessed as inhibition of kynurenine production preincubated with cells followed by IFN-g...More data for this Ligand-Target Pair
TargetIndoleamine 2,3-dioxygenase 1(Human)
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Affinity DataIC50: 540nMAssay Description:Inhibition of IDO1 in IFN-gamma stimulated human HeLa cells assessed as inhibition of kynurenine production preincubated with cells followed by IFN-g...More data for this Ligand-Target Pair
TargetIndoleamine 2,3-dioxygenase 1(Human)
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Affinity DataIC50: 570nMAssay Description:Inhibition of IDO1 in IFN-gamma stimulated human HeLa cells assessed as inhibition of kynurenine production preincubated with cells followed by IFN-g...More data for this Ligand-Target Pair
TargetIndoleamine 2,3-dioxygenase 1(Human)
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Affinity DataIC50: 690nMAssay Description:Inhibition of IDO1 in IFN-gamma stimulated human HeLa cells assessed as inhibition of kynurenine production preincubated with cells followed by IFN-g...More data for this Ligand-Target Pair
TargetIndoleamine 2,3-dioxygenase 1(Human)
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Affinity DataIC50: 790nMAssay Description:Inhibition of IDO1 in IFN-gamma stimulated human HeLa cells assessed as inhibition of kynurenine production preincubated with cells followed by IFN-g...More data for this Ligand-Target Pair
TargetIndoleamine 2,3-dioxygenase 1(Human)
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Affinity DataIC50: 830nMAssay Description:Inhibition of IDO1 in IFN-gamma stimulated human HeLa cells assessed as inhibition of kynurenine production preincubated with cells followed by IFN-g...More data for this Ligand-Target Pair
TargetIndoleamine 2,3-dioxygenase 1(Human)
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Affinity DataIC50: 840nMAssay Description:Inhibition of IDO1 in IFN-gamma stimulated human HeLa cells assessed as inhibition of kynurenine production preincubated with cells followed by IFN-g...More data for this Ligand-Target Pair
TargetIndoleamine 2,3-dioxygenase 1(Human)
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Affinity DataIC50: 1.00E+3nMAssay Description:Inhibition of IDO1 in IFN-gamma stimulated human HeLa cells assessed as inhibition of kynurenine production preincubated with cells followed by IFN-g...More data for this Ligand-Target Pair
TargetIndoleamine 2,3-dioxygenase 1(Human)
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Affinity DataIC50: 1.50E+3nMAssay Description:Inhibition of IDO1 in IFN-gamma stimulated human HeLa cells assessed as inhibition of kynurenine production preincubated with cells followed by IFN-g...More data for this Ligand-Target Pair
TargetIndoleamine 2,3-dioxygenase 1(Human)
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Affinity DataIC50: 1.80E+3nMAssay Description:Inhibition of IDO1 in IFN-gamma stimulated human HeLa cells assessed as inhibition of kynurenine production preincubated with cells followed by IFN-g...More data for this Ligand-Target Pair
TargetIndoleamine 2,3-dioxygenase 1(Human)
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Affinity DataIC50: 1.80E+3nMAssay Description:Inhibition of IDO1 in IFN-gamma stimulated human HeLa cells assessed as inhibition of kynurenine production preincubated with cells followed by IFN-g...More data for this Ligand-Target Pair
TargetIndoleamine 2,3-dioxygenase 1(Human)
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Affinity DataIC50: 2.10E+3nMAssay Description:Inhibition of IDO1 in IFN-gamma stimulated human HeLa cells assessed as inhibition of kynurenine production preincubated with cells followed by IFN-g...More data for this Ligand-Target Pair
TargetIndoleamine 2,3-dioxygenase 1(Human)
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Affinity DataIC50: 2.40E+3nMAssay Description:Inhibition of IDO1 in IFN-gamma stimulated human HeLa cells assessed as inhibition of kynurenine production preincubated with cells followed by IFN-g...More data for this Ligand-Target Pair
TargetIndoleamine 2,3-dioxygenase 1(Human)
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Affinity DataIC50: 2.40E+3nMAssay Description:Inhibition of IDO1 in IFN-gamma stimulated human HeLa cells assessed as inhibition of kynurenine production preincubated with cells followed by IFN-g...More data for this Ligand-Target Pair
Affinity DataIC50: 2.50E+3nMAssay Description:Inhibition of CYP2C9 (unknown origin)More data for this Ligand-Target Pair
TargetIndoleamine 2,3-dioxygenase 1(Human)
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Affinity DataIC50: 2.60E+3nMAssay Description:Inhibition of IDO1 in IFN-gamma stimulated human HeLa cells assessed as inhibition of kynurenine production preincubated with cells followed by IFN-g...More data for this Ligand-Target Pair
TargetIndoleamine 2,3-dioxygenase 1(Human)
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Affinity DataIC50: 2.90E+3nMAssay Description:Inhibition of IDO1 in IFN-gamma stimulated human HeLa cells assessed as inhibition of kynurenine production preincubated with cells followed by IFN-g...More data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+3nMAssay Description:Inhibition of CYP1A2 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+3nMAssay Description:Inhibition of CYP1A2 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+3nMAssay Description:Inhibition of CYP2B6 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+3nMAssay Description:Inhibition of CYP2C19 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+3nMAssay Description:Inhibition of CYP2C19 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+3nMAssay Description:Inhibition of CYP3A4 (unknown origin) using BFC as substrateMore data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+3nMAssay Description:Inhibition of CYP3A4 (unknown origin) using BFC as substrateMore data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+3nMAssay Description:Inhibition of CYP3A4 (unknown origin) using BZR as substrateMore data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+3nMAssay Description:Inhibition of CYP1A2 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+3nMAssay Description:Inhibition of CYP2D6 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+3nMAssay Description:Inhibition of CYP2C9 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+3nMAssay Description:Inhibition of CYP2C19 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+3nMAssay Description:Inhibition of CYP2D6 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+3nMAssay Description:Inhibition of CYP2B6 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+3nMAssay Description:Inhibition of CYP2D6 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+3nMAssay Description:Inhibition of CYP3A4 (unknown origin) using BZR as substrateMore data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+3nMAssay Description:Inhibition of CYP3A4 (unknown origin) using BFC as substrateMore data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+3nMAssay Description:Inhibition of CYP3A4 (unknown origin) using BZR as substrateMore data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+3nMAssay Description:Inhibition of CYP2C8 (unknown origin)More data for this Ligand-Target Pair