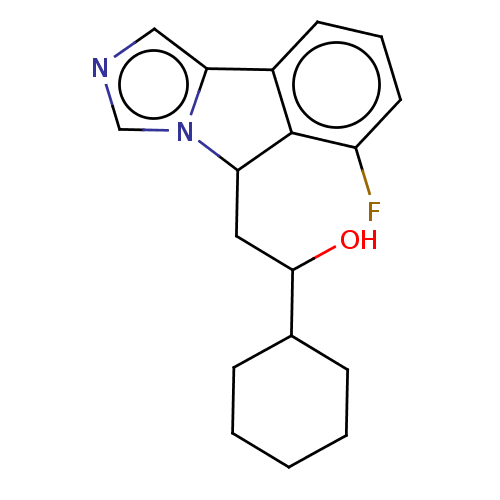
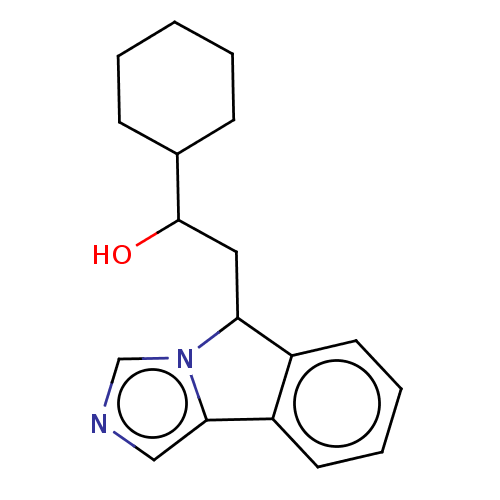
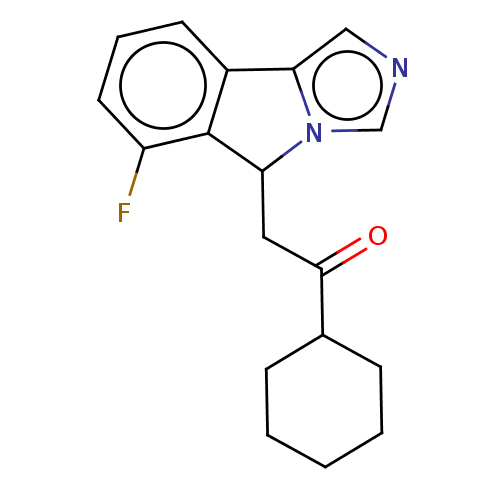
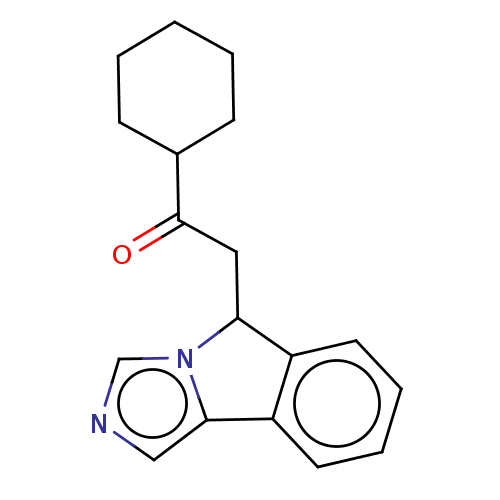
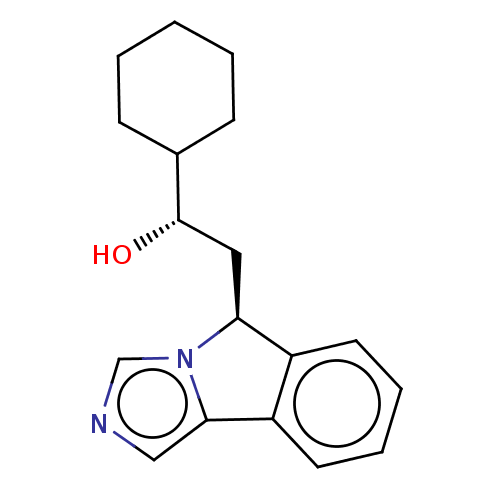
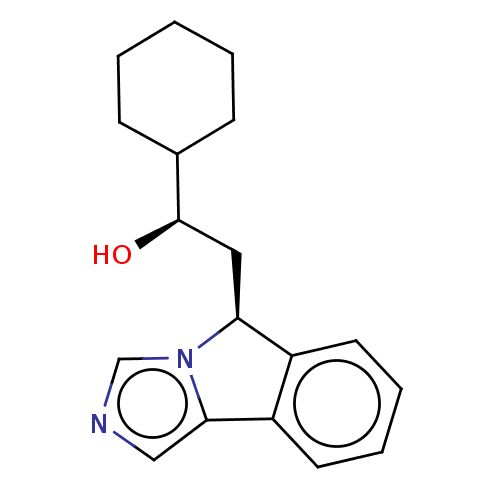
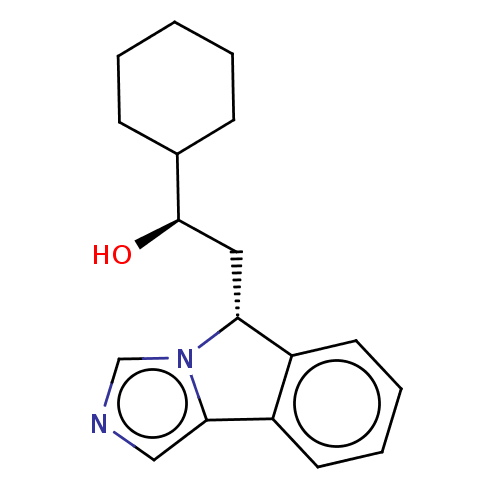
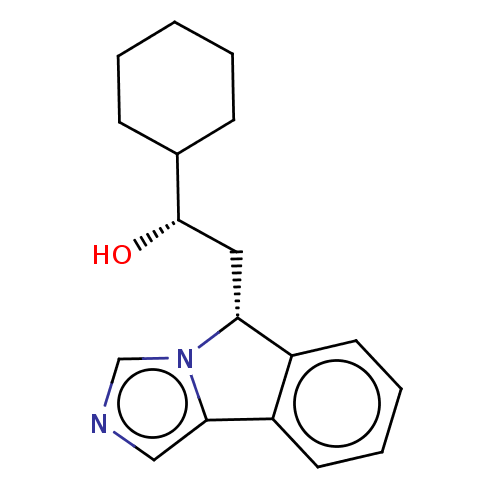
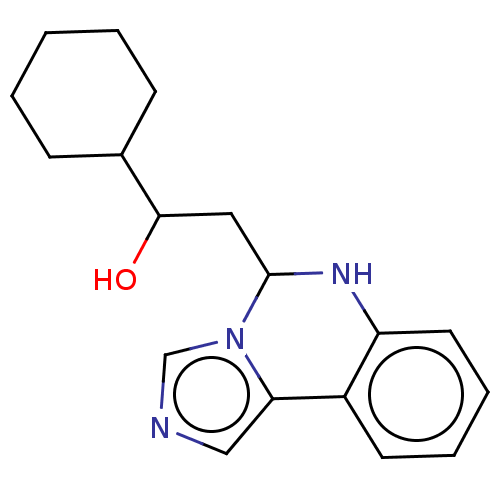
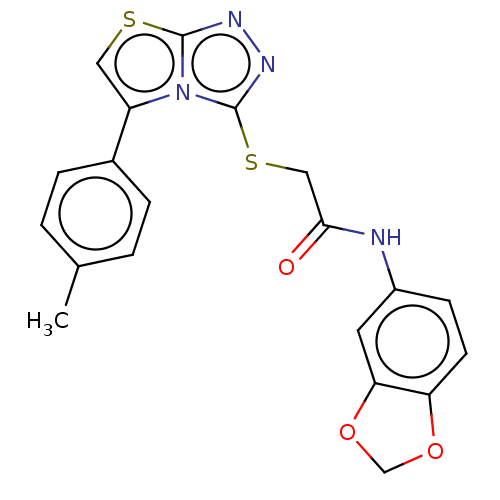
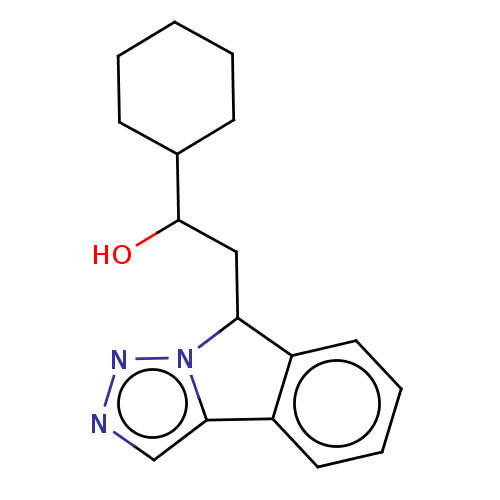
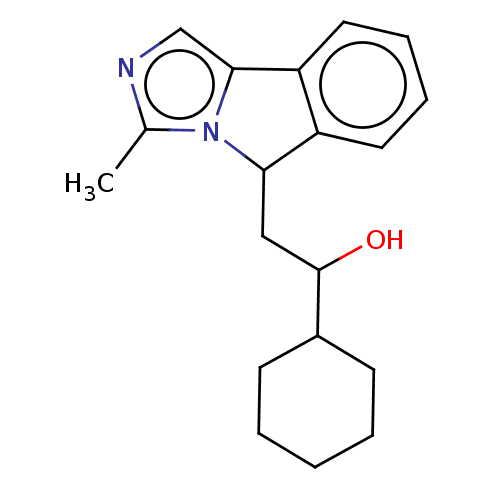
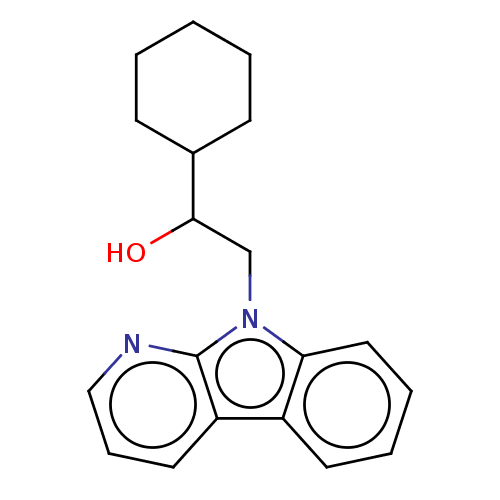
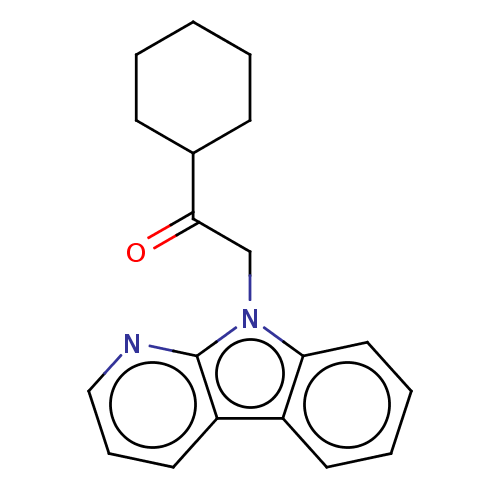
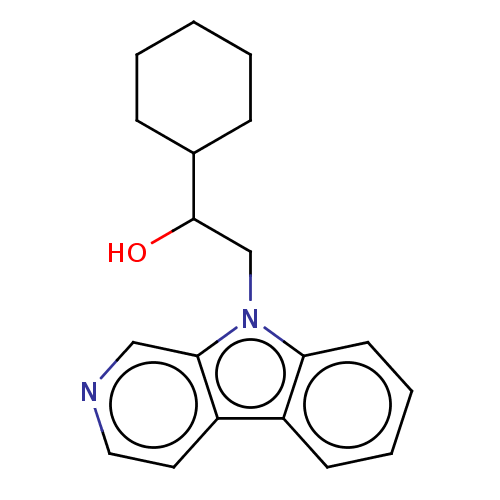
Report error Found 22 Enz. Inhib. hit(s) with all data for entry = 50046927
Affinity DataIC50: 19nMAssay Description:Inhibition of recombinant human IDO1 assessed as conversion of N-formylkynurenine to kynurenine incubated for 1 hr by fluorescence analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 38nMAssay Description:Inhibition of recombinant human IDO1 assessed as conversion of N-formylkynurenine to kynurenine incubated for 1 hr by fluorescence analysisMore data for this Ligand-Target Pair
Affinity DataEC50: 55nMAssay Description:Inhibition of IDO1 in IFNgamma-stimulated human HeLa cells incubated for 24 hrsMore data for this Ligand-Target Pair
Affinity DataEC50: 61nMAssay Description:Inhibition of IDO1 in IFNgamma-stimulated human HeLa cells incubated for 24 hrsMore data for this Ligand-Target Pair
Affinity DataIC50: 279nMAssay Description:Inhibition of recombinant human IDO1 assessed as conversion of N-formylkynurenine to kynurenine incubated for 1 hr by fluorescence analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 468nMAssay Description:Inhibition of recombinant human IDO1 assessed as conversion of N-formylkynurenine to kynurenine incubated for 1 hr by fluorescence analysisMore data for this Ligand-Target Pair
Affinity DataEC50: 725nMAssay Description:Inhibition of IDO1 in IFNgamma-stimulated human HeLa cells incubated for 24 hrsMore data for this Ligand-Target Pair
Affinity DataEC50: 749nMAssay Description:Inhibition of IDO1 in IFNgamma-stimulated human HeLa cells incubated for 24 hrsMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+3nMAssay Description:Inhibition of recombinant human IDO1 expressed in M15(pREP4) cells using L-tryptophan as substrate assessed as conversion of N-formylkynurenine to ky...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+3nMAssay Description:Inhibition of recombinant human IDO1 expressed in M15(pREP4) cells using L-tryptophan as substrate assessed as conversion of N-formylkynurenine to ky...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+3nMAssay Description:Inhibition of recombinant human IDO1 expressed in M15(pREP4) cells using L-tryptophan as substrate assessed as conversion of N-formylkynurenine to ky...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+3nMAssay Description:Inhibition of recombinant human IDO1 expressed in M15(pREP4) cells using L-tryptophan as substrate assessed as conversion of N-formylkynurenine to ky...More data for this Ligand-Target Pair
Affinity DataIC50: 1.14E+3nMAssay Description:Inhibition of recombinant human IDO1 assessed as conversion of N-formylkynurenine to kynurenine incubated for 1 hr by fluorescence analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+3nMAssay Description:Inhibition of human IDO1 by Bridge-IT Tryptophan fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibition of recombinant human IDO1 assessed as conversion of N-formylkynurenine to kynurenine incubated for 1 hr by fluorescence analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibition of recombinant human IDO1 assessed as conversion of N-formylkynurenine to kynurenine incubated for 1 hr by fluorescence analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibition of recombinant human IDO1 assessed as conversion of N-formylkynurenine to kynurenine incubated for 1 hr by fluorescence analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibition of recombinant human IDO1 assessed as conversion of N-formylkynurenine to kynurenine incubated for 1 hr by fluorescence analysisMore data for this Ligand-Target Pair
Affinity DataEC50: >1.00E+4nMAssay Description:Inhibition of IDO1 in IFNgamma-stimulated human HeLa cells incubated for 24 hrsMore data for this Ligand-Target Pair
Affinity DataEC50: >1.00E+4nMAssay Description:Inhibition of IDO1 in IFNgamma-stimulated human HeLa cells incubated for 24 hrsMore data for this Ligand-Target Pair
Affinity DataEC50: >1.00E+4nMAssay Description:Inhibition of IDO1 in IFNgamma-stimulated human HeLa cells incubated for 24 hrsMore data for this Ligand-Target Pair
Affinity DataIC50: 4.80E+4nMAssay Description:Inhibition of recombinant human IDO1 assessed as conversion of N-formylkynurenine to kynurenine incubated for 1 hr by fluorescence analysisMore data for this Ligand-Target Pair