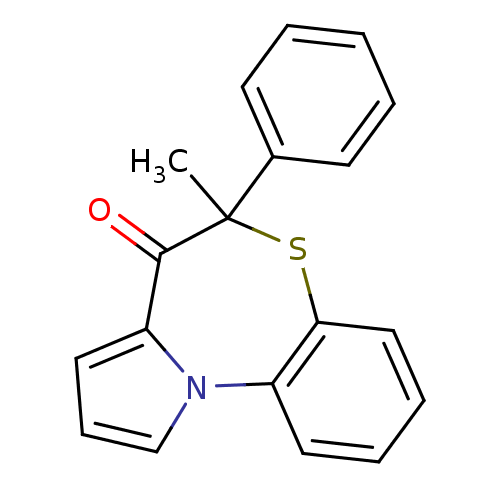
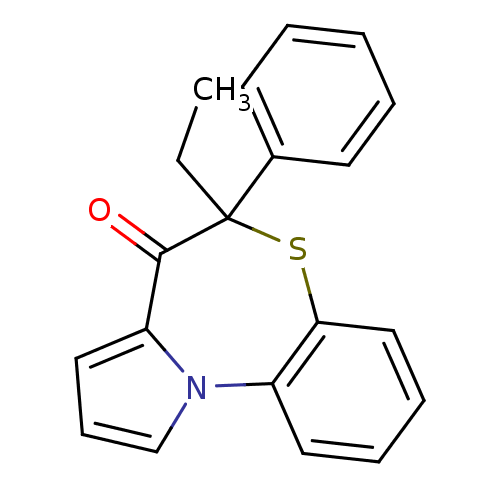
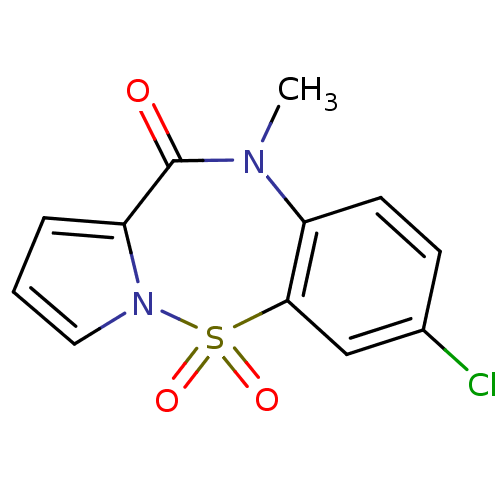
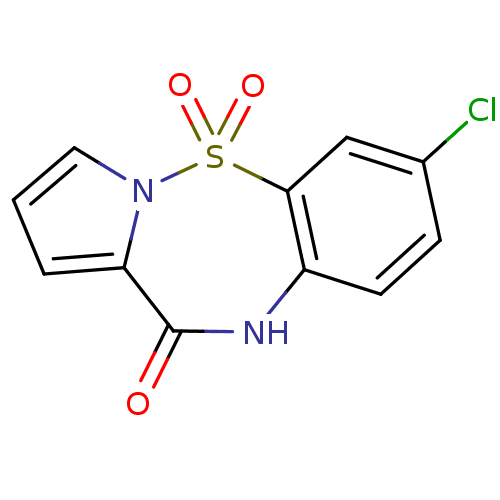
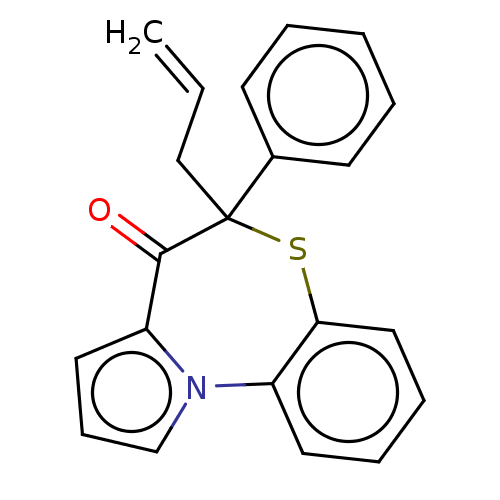
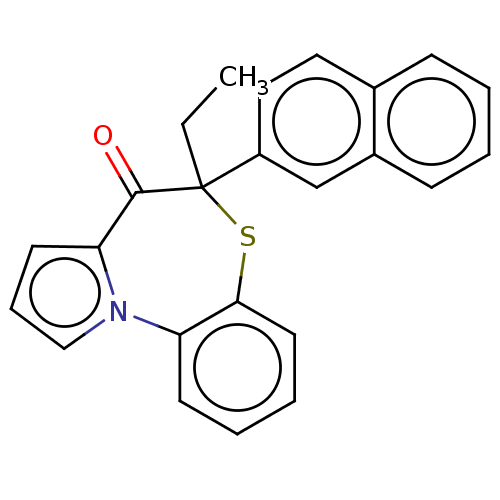
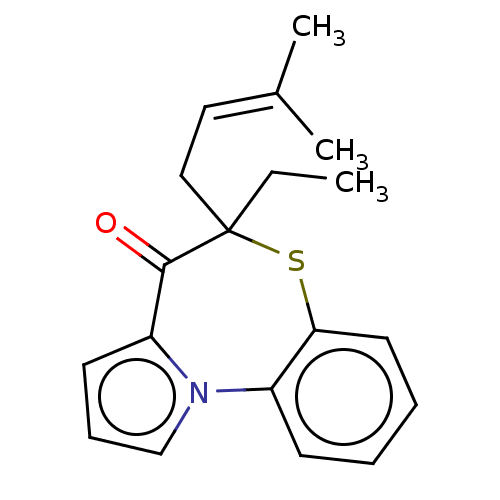
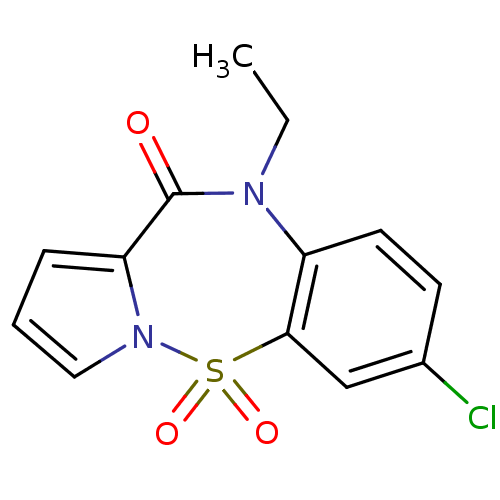
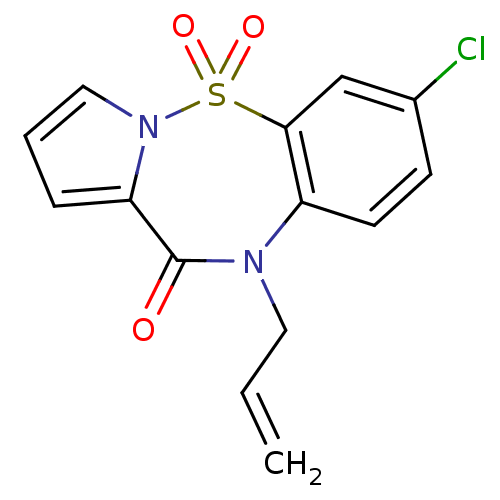
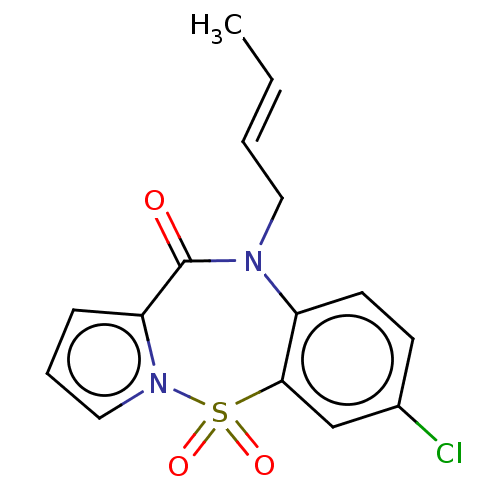
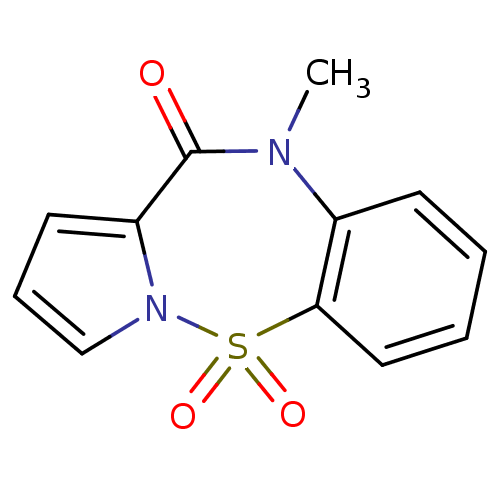
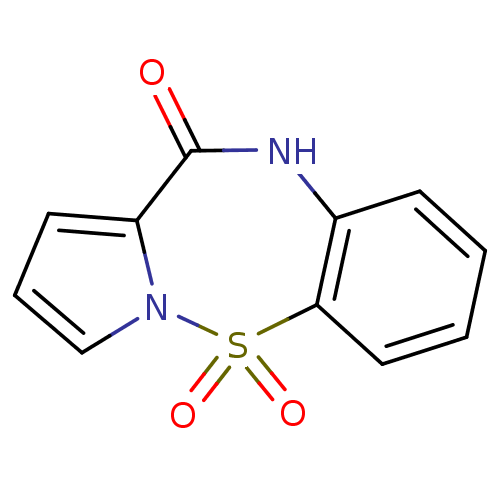
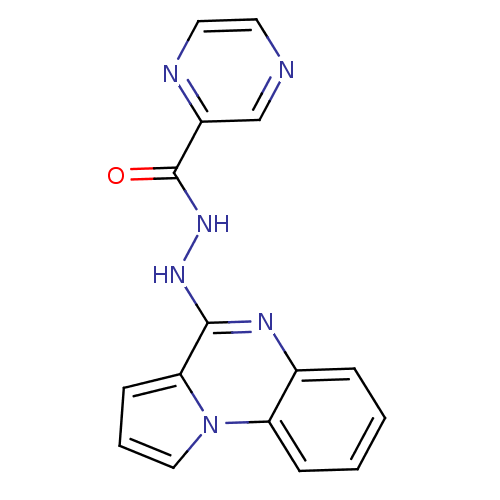
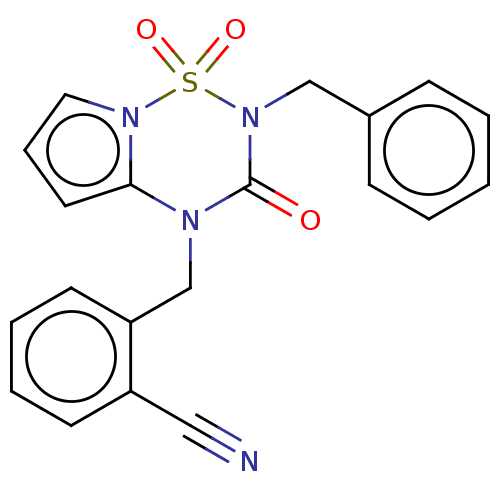
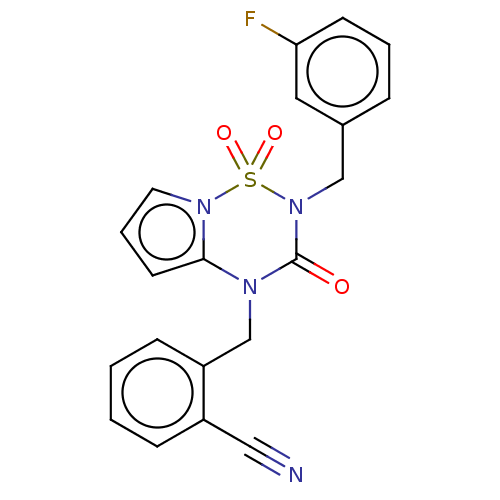
Report error Found 19 Enz. Inhib. hit(s) with all data for entry = 50006294
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
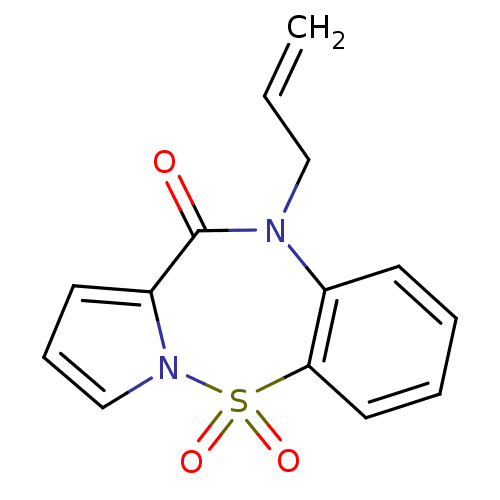
Affinity DataIC50: 350nMAssay Description:Inhibition of HIV1 reverse transcriptaseMore data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
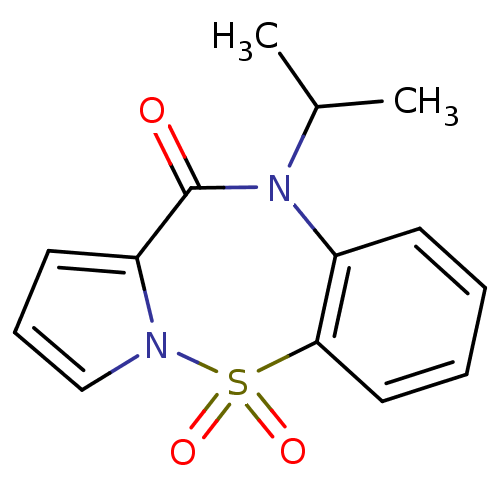
Affinity DataIC50: 470nMAssay Description:Inhibition of HIV1 reverse transcriptaseMore data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
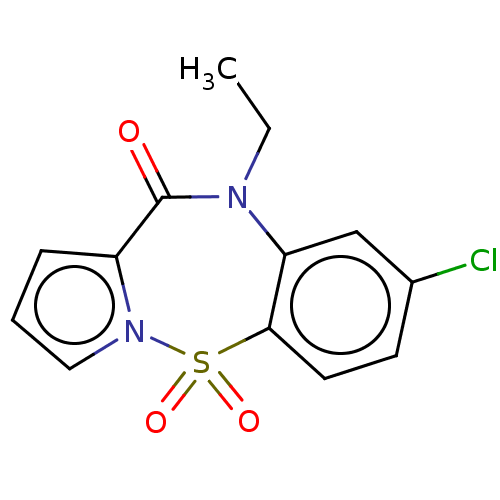
Affinity DataIC50: 500nMAssay Description:Inhibition of HIV1 reverse transcriptaseMore data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
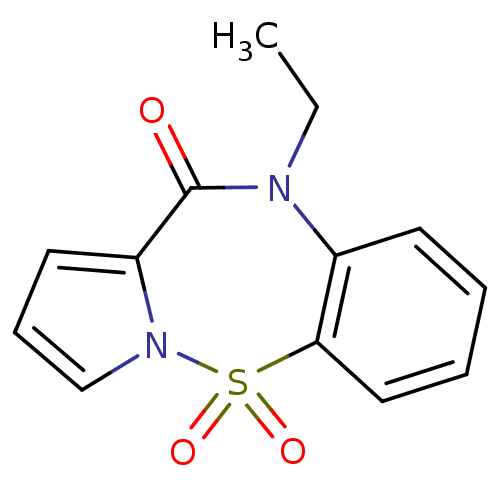
Affinity DataIC50: 700nMAssay Description:Inhibition of HIV1 reverse transcriptaseMore data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
Affinity DataIC50: 710nMAssay Description:Inhibition of HIV1 reverse transcriptaseMore data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
Affinity DataIC50: 850nMAssay Description:Inhibition of HIV1 reverse transcriptaseMore data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
Affinity DataIC50: 1.35E+3nMAssay Description:Inhibition of HIV1 reverse transcriptaseMore data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
Affinity DataIC50: 2.70E+3nMAssay Description:Inhibition of HIV1 reverse transcriptaseMore data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
Affinity DataIC50: 2.90E+3nMAssay Description:Inhibition of HIV1 reverse transcriptaseMore data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
Affinity DataIC50: 3.10E+3nMAssay Description:Inhibition of HIV1 reverse transcriptaseMore data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
Affinity DataIC50: 4.00E+3nMAssay Description:Inhibition of HIV1 reverse transcriptaseMore data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
Affinity DataIC50: 4.00E+3nMAssay Description:Inhibition of HIV1 reverse transcriptaseMore data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
Affinity DataIC50: 4.00E+3nMAssay Description:Inhibition of HIV1 reverse transcriptaseMore data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
Affinity DataIC50: 4.00E+3nMAssay Description:Inhibition of HIV1 reverse transcriptaseMore data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
Affinity DataIC50: 4.00E+3nMAssay Description:Inhibition of HIV1 reverse transcriptaseMore data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
Affinity DataIC50: 4.00E+3nMAssay Description:Inhibition of HIV1 reverse transcriptaseMore data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
Affinity DataIC50: 1.90E+4nMAssay Description:Inhibition of HIV1 reverse transcriptaseMore data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
Affinity DataIC50: 5.69E+4nMAssay Description:Inhibition of HIV1 reverse transcriptase assessed as inhibition of biotin-dUTP incorporationMore data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
Institute of Experimental Botany Ascr & Palack£
Curated by ChEMBL
Affinity DataIC50: 5.83E+4nMAssay Description:Inhibition of HIV1 reverse transcriptase assessed as inhibition of biotin-dUTP incorporationMore data for this Ligand-Target Pair