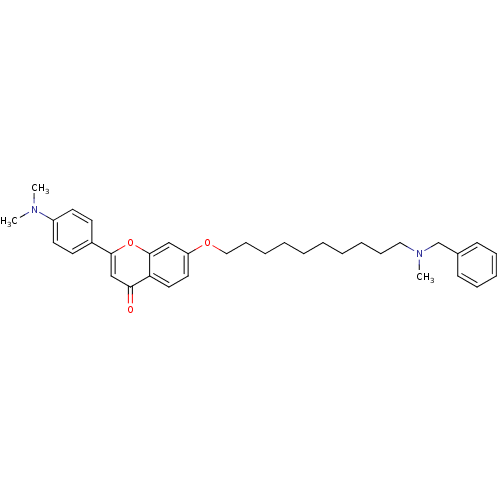
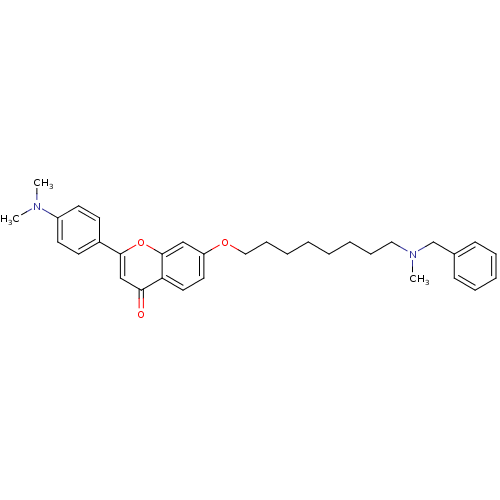
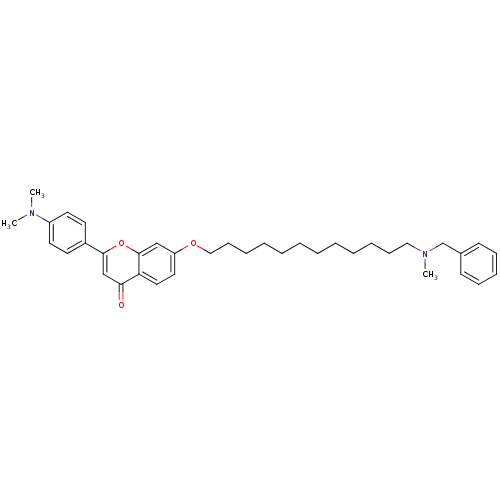
Report error Found 41 Enz. Inhib. hit(s) with all data for entry = 50043621
Affinity DataIC50: 47nMAssay Description:Inhibition of equine serum BChE using butylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 219nMAssay Description:Inhibition of electric eel AChE using acetylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataKi: 386nMAssay Description:Mixed-type inhibition of electric eel AChE using acetylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Linewea...More data for this Ligand-Target Pair
Affinity DataIC50: 820nMAssay Description:Inhibition of equine serum BChE using butylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 1.71E+3nMAssay Description:Inhibition of equine serum BChE using butylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 1.83E+3nMAssay Description:Inhibition of electric eel AChE using acetylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 1.99E+3nMAssay Description:Inhibition of electric eel AChE using acetylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 2.24E+3nMAssay Description:Inhibition of electric eel AChE using acetylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 2.26E+3nMAssay Description:Inhibition of electric eel AChE using acetylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 2.31E+3nMAssay Description:Inhibition of equine serum BChE using butylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 2.42E+3nMAssay Description:Inhibition of equine serum BChE using butylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 2.60E+3nMAssay Description:Inhibition of equine serum BChE using butylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 2.77E+3nMAssay Description:Inhibition of electric eel AChE using acetylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 3.04E+3nMAssay Description:Inhibition of electric eel AChE using acetylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 3.11E+3nMAssay Description:Inhibition of electric eel AChE using acetylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 3.74E+3nMAssay Description:Inhibition of electric eel AChE using acetylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 4.61E+3nMAssay Description:Inhibition of equine serum BChE using butylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 5.01E+3nMAssay Description:Inhibition of equine serum BChE using butylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 5.52E+3nMAssay Description:Inhibition of equine serum BChE using butylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 5.56E+3nMAssay Description:Inhibition of electric eel AChE using acetylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 6.12E+3nMAssay Description:Inhibition of equine serum BChE using butylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 6.14E+3nMAssay Description:Inhibition of equine serum BChE using butylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 6.73E+3nMAssay Description:Inhibition of electric eel AChE using acetylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 7.85E+3nMAssay Description:Inhibition of electric eel AChE using acetylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 7.88E+3nMAssay Description:Inhibition of equine serum BChE using butylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 7.88E+3nMAssay Description:Inhibition of electric eel AChE using acetylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 8.29E+3nMAssay Description:Inhibition of electric eel AChE using acetylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 8.72E+3nMAssay Description:Inhibition of equine serum BChE using butylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 9.18E+3nMAssay Description:Inhibition of electric eel AChE using acetylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 1.01E+4nMAssay Description:Inhibition of equine serum BChE using butylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 1.14E+4nMAssay Description:Inhibition of electric eel AChE using acetylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 1.15E+4nMAssay Description:Inhibition of equine serum BChE using butylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 2.40E+4nMAssay Description:Inhibition of electric eel AChE using acetylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 3.32E+4nMAssay Description:Inhibition of electric eel AChE using acetylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:Inhibition of equine serum BChE using butylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:Inhibition of electric eel AChE using acetylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:Inhibition of equine serum BChE using butylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:Inhibition of equine serum BChE using butylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:Inhibition of electric eel AChE using acetylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:Inhibition of equine serum BChE using butylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:Inhibition of equine serum BChE using butylthiocholine chloride as substrate incubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
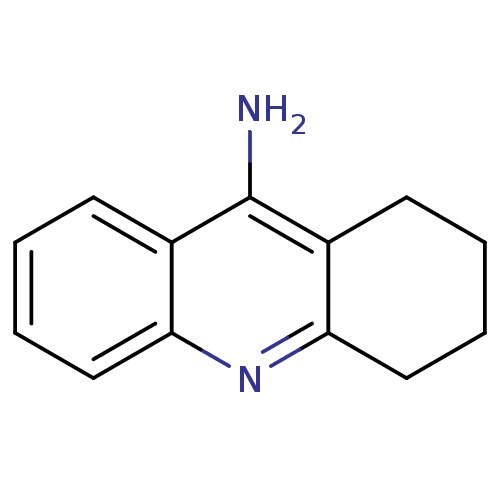
 3D Structure (crystal)
3D Structure (crystal)