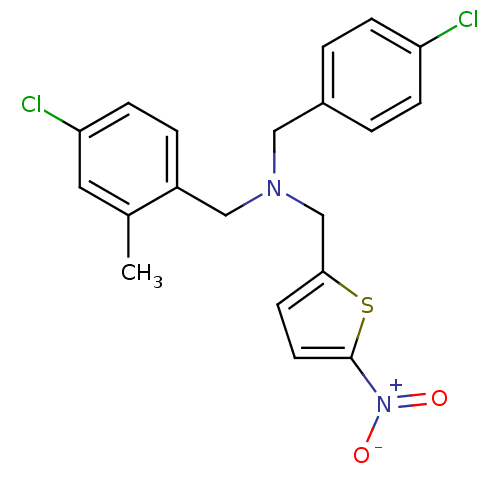
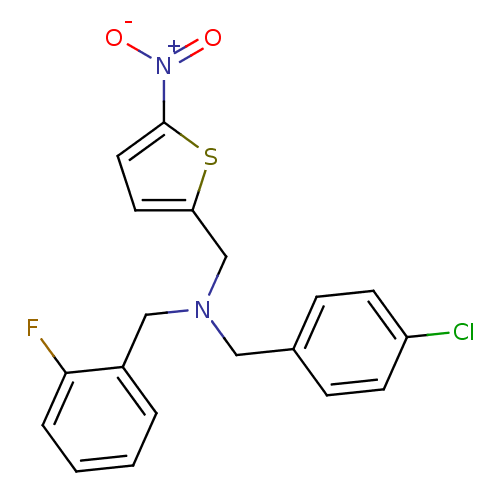
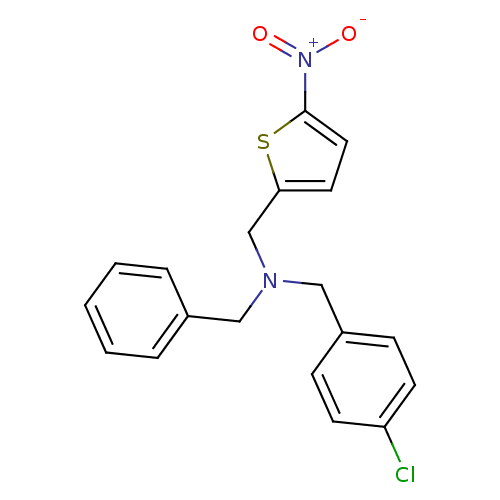
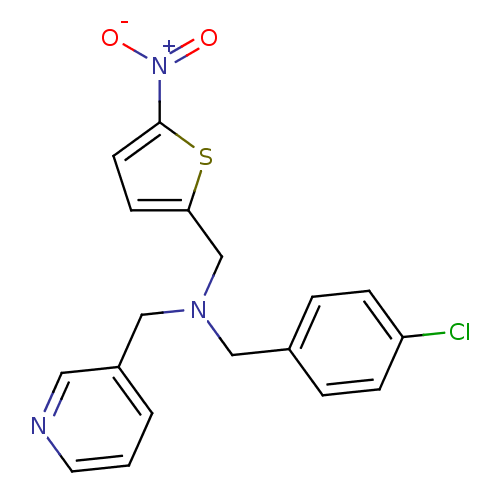
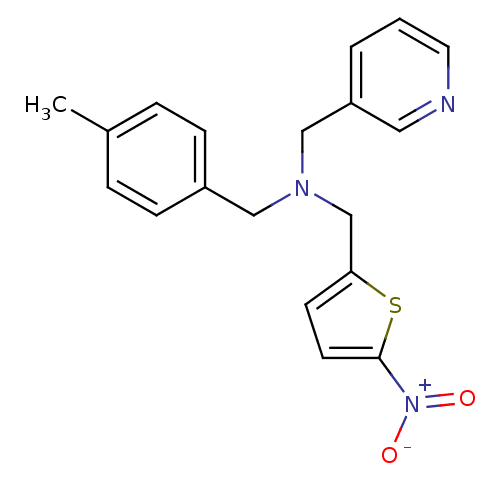
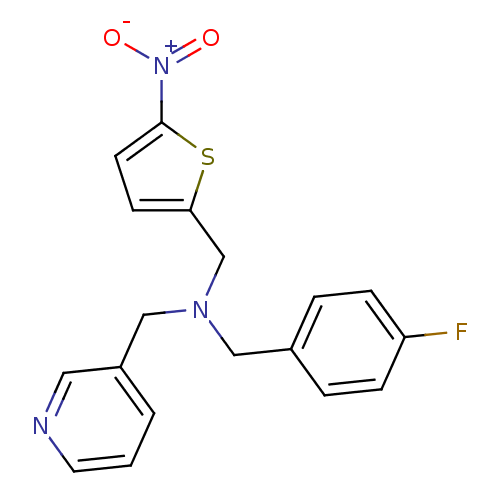
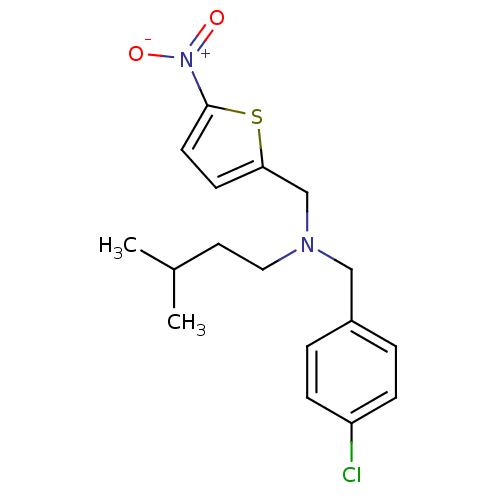
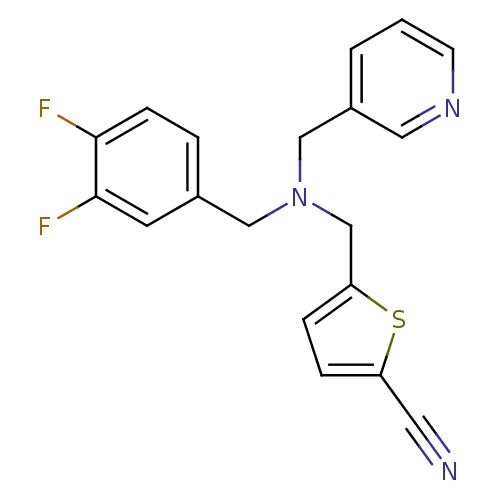
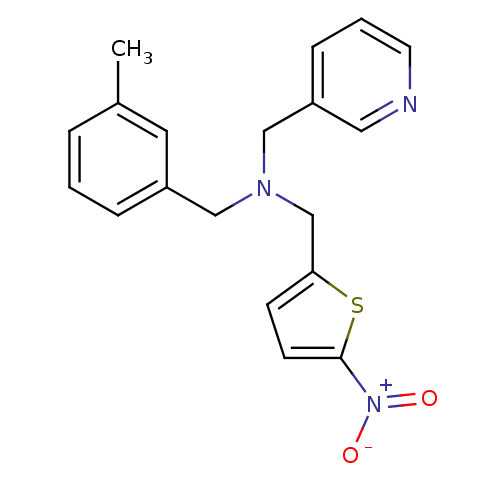
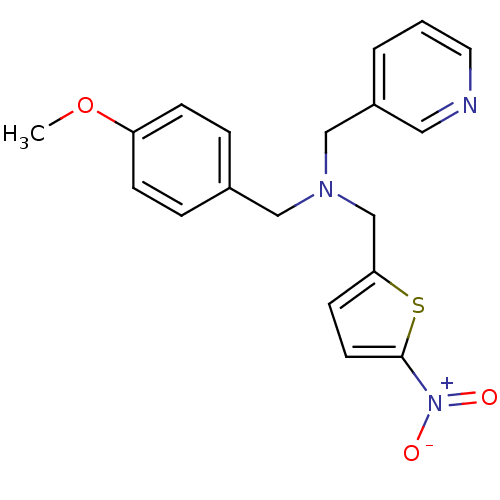
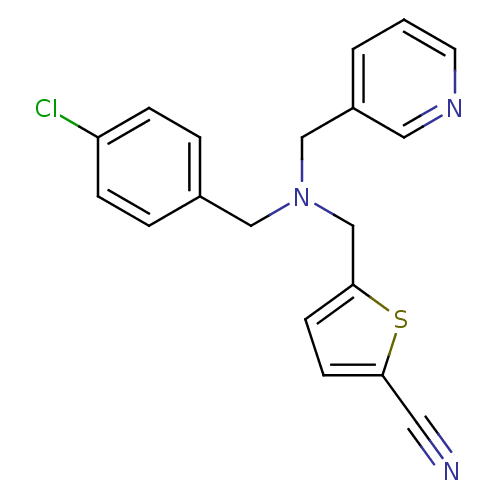
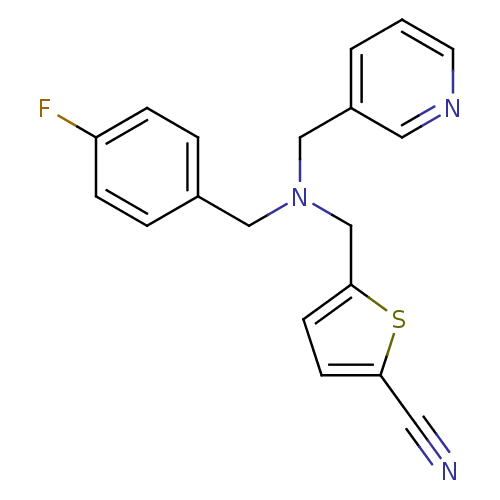
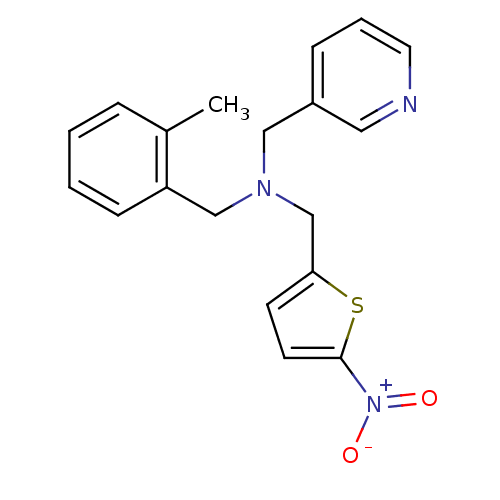
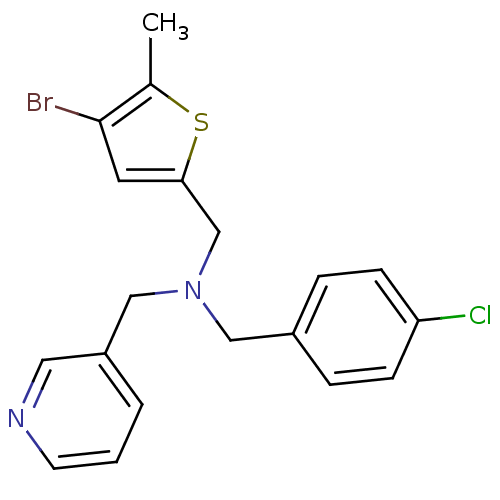
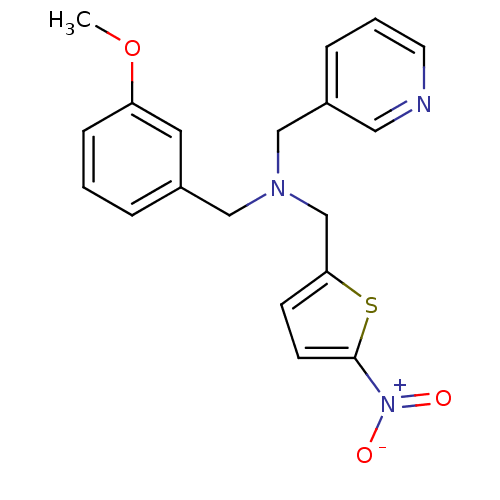
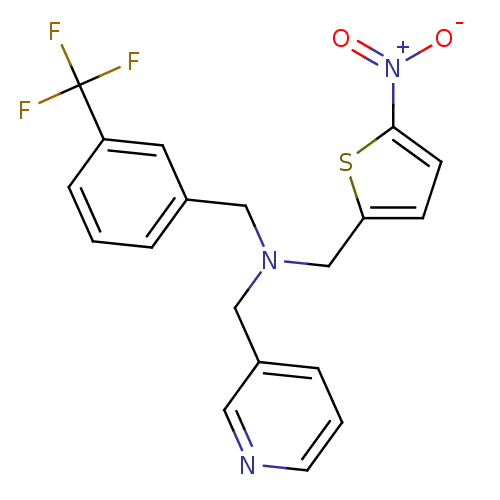
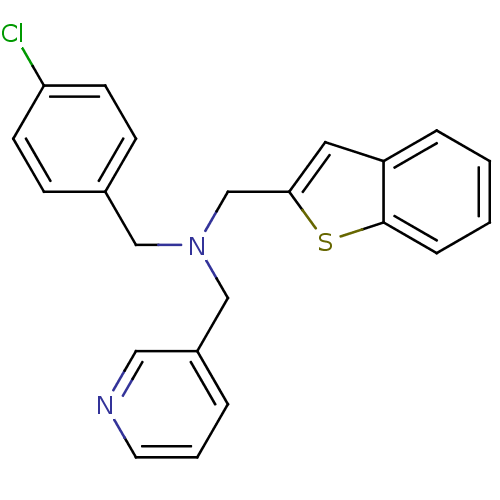
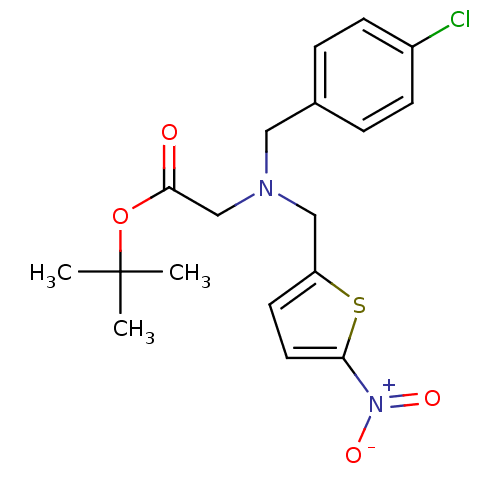
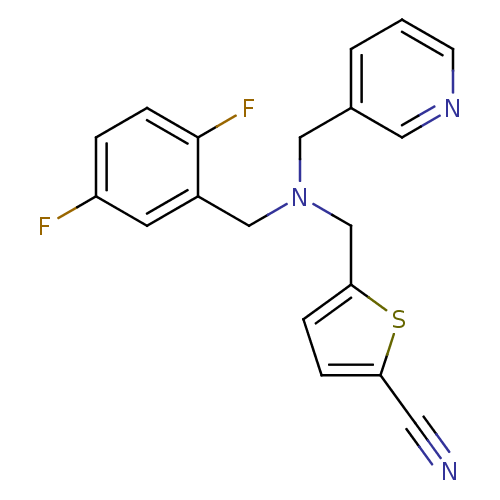
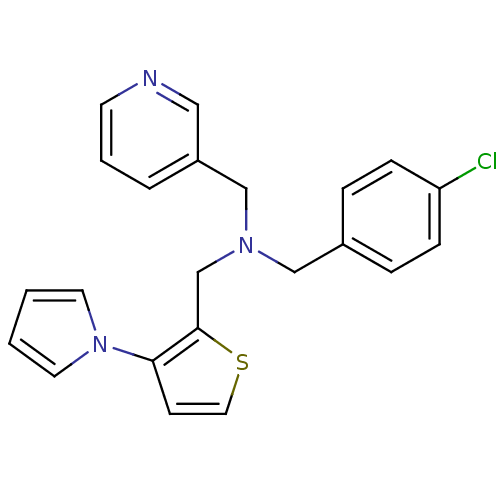
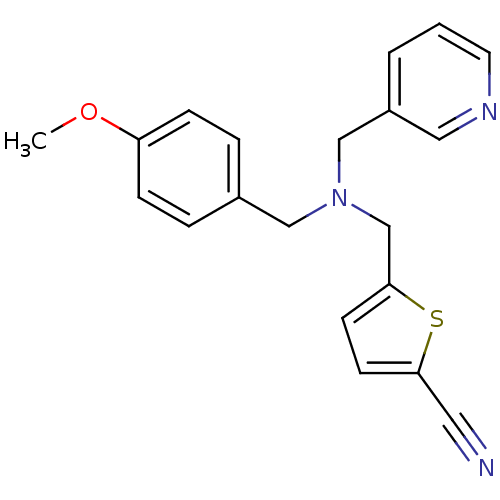
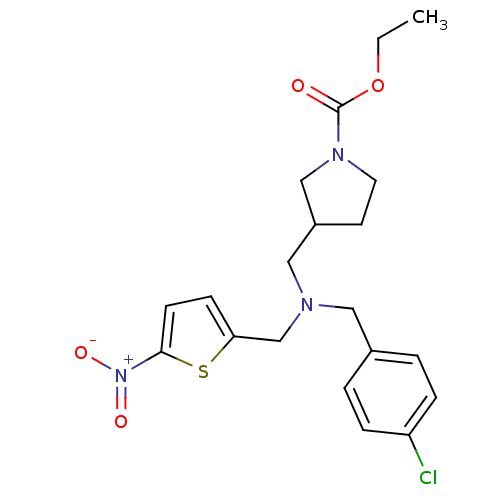
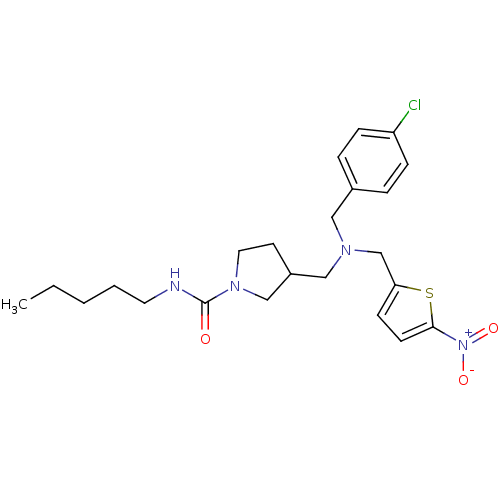
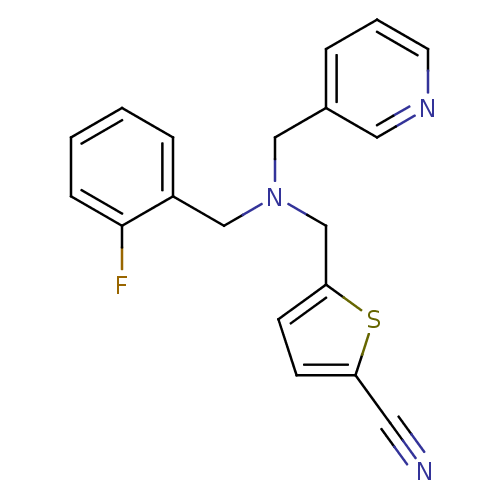
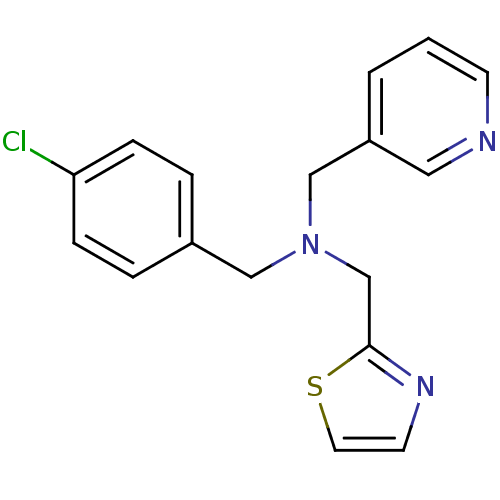
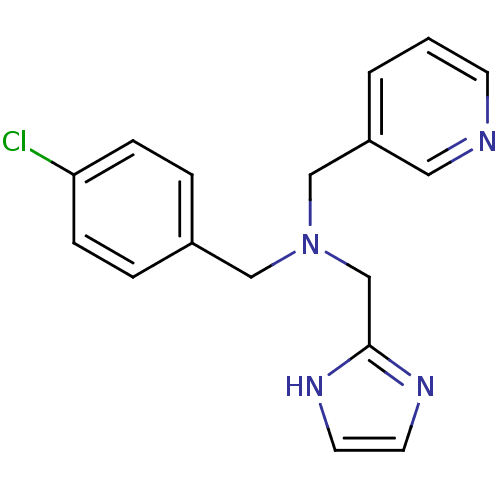
Report error Found 51 Enz. Inhib. hit(s) with all data for entry = 50042972
Affinity DataEC50: 50nMAssay Description:Agonist activity at biotinylated REV-ERBalpha (unknown origin) assessed as increase in biotinylated NCOR peptide recruitment after 1 hr by FRET assayMore data for this Ligand-Target Pair
Affinity DataEC50: 63nMAssay Description:Agonist activity at biotinylated REV-ERBalpha (unknown origin) assessed as increase in biotinylated NCOR peptide recruitment after 1 hr by FRET assayMore data for this Ligand-Target Pair
Affinity DataEC50: 100nMAssay Description:Agonist activity at biotinylated REV-ERBalpha (unknown origin) assessed as increase in biotinylated NCOR peptide recruitment after 1 hr by FRET assayMore data for this Ligand-Target Pair
Affinity DataEC50: 160nMAssay Description:Agonist activity at biotinylated REV-ERBalpha (unknown origin) assessed as increase in biotinylated NCOR peptide recruitment after 1 hr by FRET assayMore data for this Ligand-Target Pair
Affinity DataEC50: 160nMAssay Description:Agonist activity at biotinylated REV-ERBalpha (unknown origin) assessed as increase in biotinylated NCOR peptide recruitment after 1 hr by FRET assayMore data for this Ligand-Target Pair
Affinity DataEC50: 160nMAssay Description:Agonist activity at biotinylated REV-ERBalpha (unknown origin) assessed as increase in biotinylated NCOR peptide recruitment after 1 hr by FRET assayMore data for this Ligand-Target Pair
Affinity DataEC50: 160nMAssay Description:Agonist activity at biotinylated REV-ERBalpha (unknown origin) assessed as increase in biotinylated NCOR peptide recruitment after 1 hr by FRET assayMore data for this Ligand-Target Pair
Affinity DataEC50: 200nMAssay Description:Agonist activity at biotinylated REV-ERBalpha (unknown origin) assessed as increase in biotinylated NCOR peptide recruitment after 1 hr by FRET assayMore data for this Ligand-Target Pair
Affinity DataEC50: 200nMAssay Description:Agonist activity at biotinylated REV-ERBalpha (unknown origin) assessed as increase in biotinylated NCOR peptide recruitment after 1 hr by FRET assayMore data for this Ligand-Target Pair
Affinity DataEC50: 200nMAssay Description:Agonist activity at biotinylated REV-ERBalpha (unknown origin) assessed as increase in biotinylated NCOR peptide recruitment after 1 hr by FRET assayMore data for this Ligand-Target Pair
Affinity DataEC50: 200nMAssay Description:Agonist activity at biotinylated REV-ERBalpha (unknown origin) assessed as increase in biotinylated NCOR peptide recruitment after 1 hr by FRET assayMore data for this Ligand-Target Pair
Affinity DataEC50: 200nMAssay Description:Agonist activity at biotinylated REV-ERBalpha (unknown origin) assessed as increase in biotinylated NCOR peptide recruitment after 1 hr by FRET assayMore data for this Ligand-Target Pair
Affinity DataEC50: 200nMAssay Description:Agonist activity at biotinylated REV-ERBalpha (unknown origin) assessed as increase in biotinylated NCOR peptide recruitment after 1 hr by FRET assayMore data for this Ligand-Target Pair
Affinity DataEC50: 250nMAssay Description:Agonist activity at biotinylated REV-ERBalpha (unknown origin) assessed as increase in biotinylated NCOR peptide recruitment after 1 hr by FRET assayMore data for this Ligand-Target Pair
Affinity DataEC50: 400nMAssay Description:Agonist activity at biotinylated REV-ERBalpha (unknown origin) assessed as increase in biotinylated NCOR peptide recruitment after 1 hr by FRET assayMore data for this Ligand-Target Pair
Affinity DataEC50: 400nMAssay Description:Agonist activity at biotinylated REV-ERBalpha (unknown origin) assessed as increase in biotinylated NCOR peptide recruitment after 1 hr by FRET assayMore data for this Ligand-Target Pair
Affinity DataEC50: 400nMAssay Description:Agonist activity at biotinylated REV-ERBalpha (unknown origin) assessed as increase in biotinylated NCOR peptide recruitment after 1 hr by FRET assayMore data for this Ligand-Target Pair
Affinity DataEC50: 500nMAssay Description:Agonist activity at biotinylated REV-ERBalpha (unknown origin) assessed as increase in biotinylated NCOR peptide recruitment after 1 hr by FRET assayMore data for this Ligand-Target Pair
Affinity DataEC50: 630nMAssay Description:Agonist activity at biotinylated REV-ERBalpha (unknown origin) assessed as increase in biotinylated NCOR peptide recruitment after 1 hr by FRET assayMore data for this Ligand-Target Pair
Affinity DataEC50: 630nMAssay Description:Agonist activity at biotinylated REV-ERBalpha (unknown origin) assessed as increase in biotinylated NCOR peptide recruitment after 1 hr by FRET assayMore data for this Ligand-Target Pair
Affinity DataEC50: 630nMAssay Description:Agonist activity at biotinylated REV-ERBalpha (unknown origin) assessed as increase in biotinylated NCOR peptide recruitment after 1 hr by FRET assayMore data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+3nMAssay Description:Binding affinity to LXRalpha (unknown origin) by radioligand displacement assayMore data for this Ligand-Target Pair
Affinity DataIC50: 6.30E+3nMAssay Description:Binding affinity to LXRalpha (unknown origin) by radioligand displacement assayMore data for this Ligand-Target Pair
Affinity DataIC50: 6.30E+3nMAssay Description:Binding affinity to LXRalpha (unknown origin) by radioligand displacement assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Binding affinity to LXRalpha (unknown origin) by radioligand displacement assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.30E+4nMAssay Description:Binding affinity to LXRalpha (unknown origin) by radioligand displacement assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.30E+4nMAssay Description:Binding affinity to LXRalpha (unknown origin) by radioligand displacement assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.60E+4nMAssay Description:Binding affinity to LXRalpha (unknown origin) by radioligand displacement assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.60E+4nMAssay Description:Binding affinity to LXRalpha (unknown origin) by radioligand displacement assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.60E+4nMAssay Description:Binding affinity to LXRalpha (unknown origin) by radioligand displacement assayMore data for this Ligand-Target Pair
Affinity DataIC50: 2.00E+4nMAssay Description:Binding affinity to LXRalpha (unknown origin) by radioligand displacement assayMore data for this Ligand-Target Pair
Affinity DataIC50: 2.00E+4nMAssay Description:Binding affinity to LXRalpha (unknown origin) by radioligand displacement assayMore data for this Ligand-Target Pair
Affinity DataIC50: 2.50E+4nMAssay Description:Binding affinity to LXRalpha (unknown origin) by radioligand displacement assayMore data for this Ligand-Target Pair
Affinity DataIC50: 3.20E+4nMAssay Description:Binding affinity to LXRalpha (unknown origin) by radioligand displacement assayMore data for this Ligand-Target Pair
Affinity DataIC50: 3.20E+4nMAssay Description:Binding affinity to LXRalpha (unknown origin) by radioligand displacement assayMore data for this Ligand-Target Pair
Affinity DataIC50: 3.20E+4nMAssay Description:Binding affinity to LXRalpha (unknown origin) by radioligand displacement assayMore data for this Ligand-Target Pair
Affinity DataIC50: 3.20E+4nMAssay Description:Binding affinity to LXRalpha (unknown origin) by radioligand displacement assayMore data for this Ligand-Target Pair
Affinity DataIC50: 4.00E+4nMAssay Description:Binding affinity to LXRalpha (unknown origin) by radioligand displacement assayMore data for this Ligand-Target Pair
Affinity DataIC50: 4.00E+4nMAssay Description:Binding affinity to LXRalpha (unknown origin) by radioligand displacement assayMore data for this Ligand-Target Pair
Affinity DataIC50: 4.00E+4nMAssay Description:Binding affinity to LXRalpha (unknown origin) by radioligand displacement assayMore data for this Ligand-Target Pair
Affinity DataIC50: 4.00E+4nMAssay Description:Binding affinity to LXRalpha (unknown origin) by radioligand displacement assayMore data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:Binding affinity to LXRalpha (unknown origin) by radioligand displacement assayMore data for this Ligand-Target Pair
Affinity DataEC50: >5.00E+4nMAssay Description:Agonist activity at biotinylated REV-ERBalpha (unknown origin) assessed as increase in biotinylated NCOR peptide recruitment after 1 hr by FRET assayMore data for this Ligand-Target Pair
Affinity DataEC50: >5.00E+4nMAssay Description:Agonist activity at biotinylated REV-ERBalpha (unknown origin) assessed as increase in biotinylated NCOR peptide recruitment after 1 hr by FRET assayMore data for this Ligand-Target Pair
Affinity DataEC50: >5.00E+4nMAssay Description:Agonist activity at biotinylated REV-ERBalpha (unknown origin) assessed as increase in biotinylated NCOR peptide recruitment after 1 hr by FRET assayMore data for this Ligand-Target Pair
Affinity DataEC50: >5.00E+4nMAssay Description:Agonist activity at biotinylated REV-ERBalpha (unknown origin) assessed as increase in biotinylated NCOR peptide recruitment after 1 hr by FRET assayMore data for this Ligand-Target Pair
Affinity DataEC50: >5.00E+4nMAssay Description:Agonist activity at biotinylated REV-ERBalpha (unknown origin) assessed as increase in biotinylated NCOR peptide recruitment after 1 hr by FRET assayMore data for this Ligand-Target Pair
Affinity DataIC50: 6.30E+4nMAssay Description:Binding affinity to LXRalpha (unknown origin) by radioligand displacement assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+5nMAssay Description:Binding affinity to LXRalpha (unknown origin) by radioligand displacement assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+5nMAssay Description:Binding affinity to LXRalpha (unknown origin) by radioligand displacement assayMore data for this Ligand-Target Pair