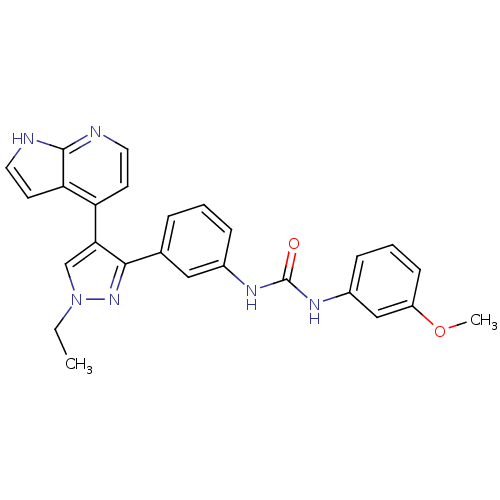
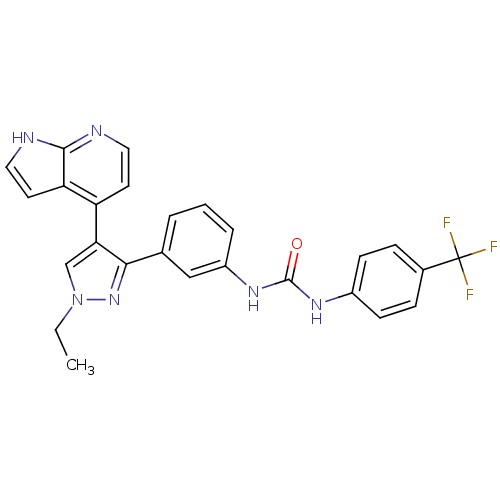
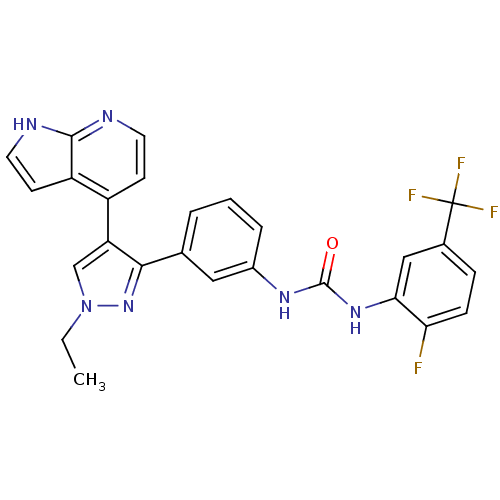
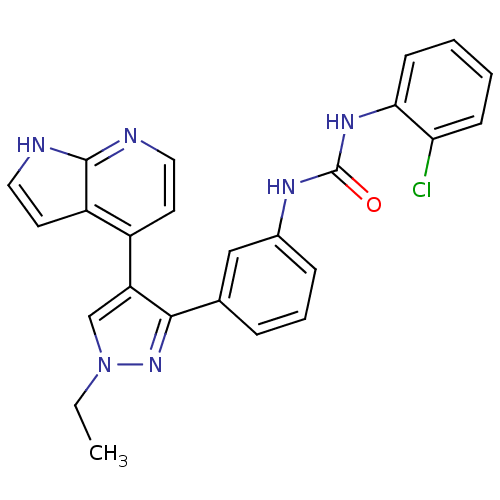
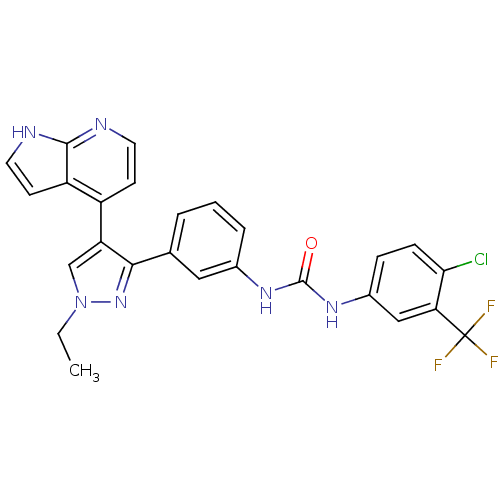
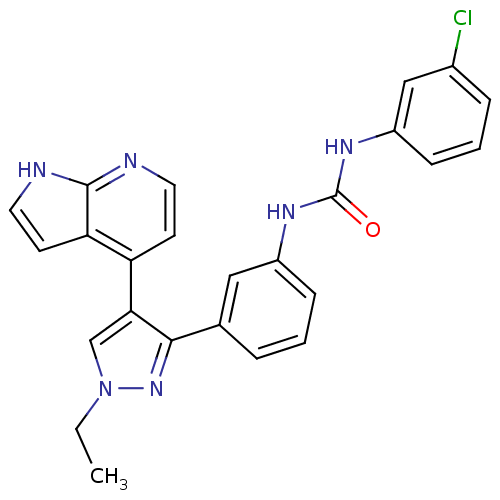
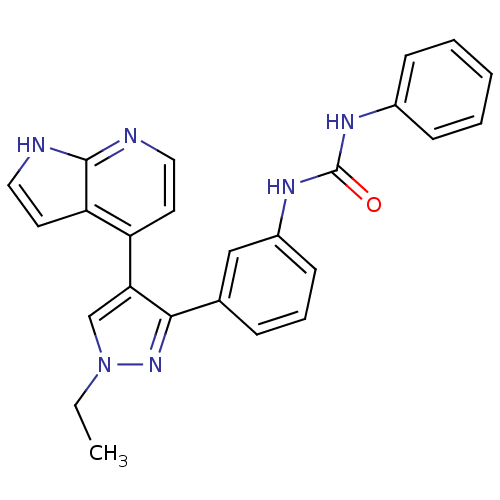
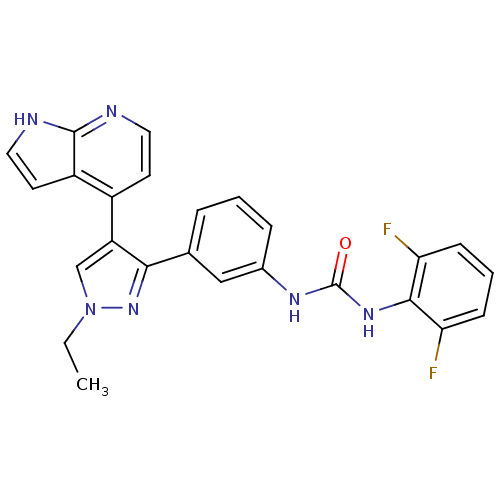
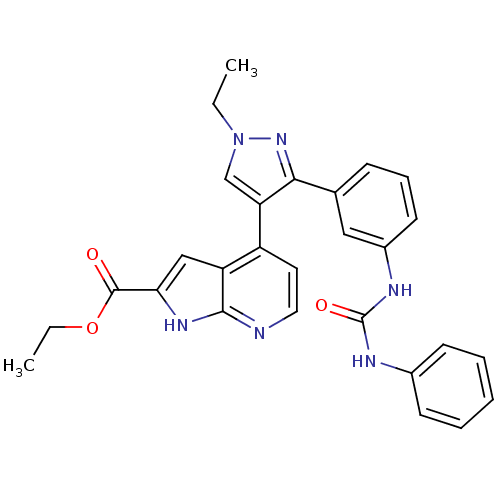
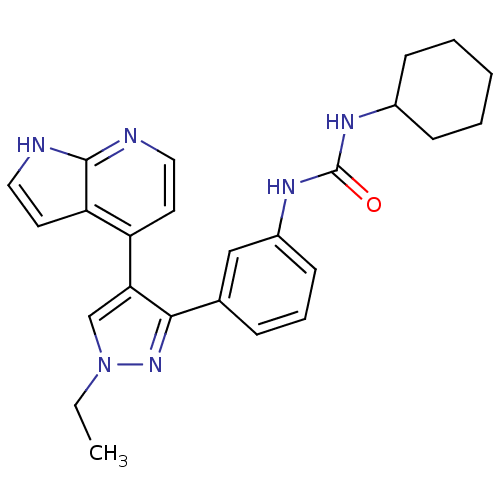
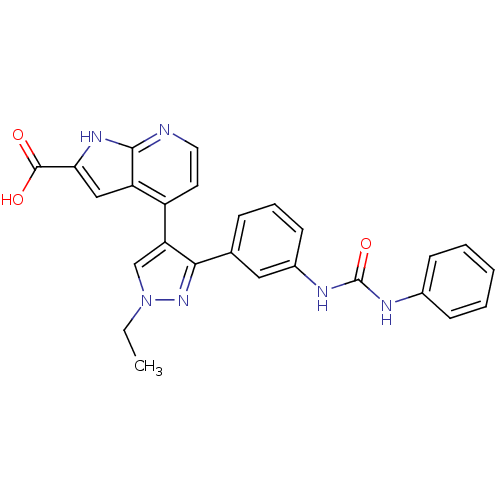
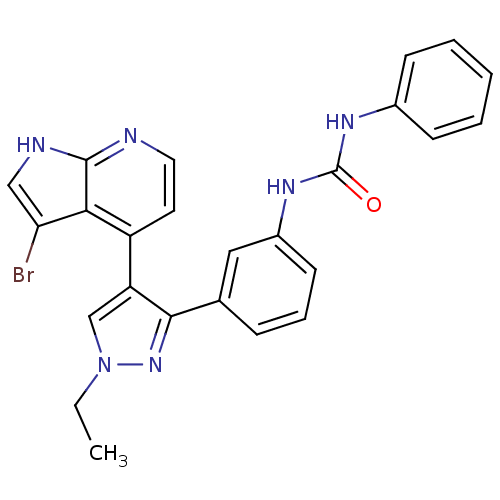
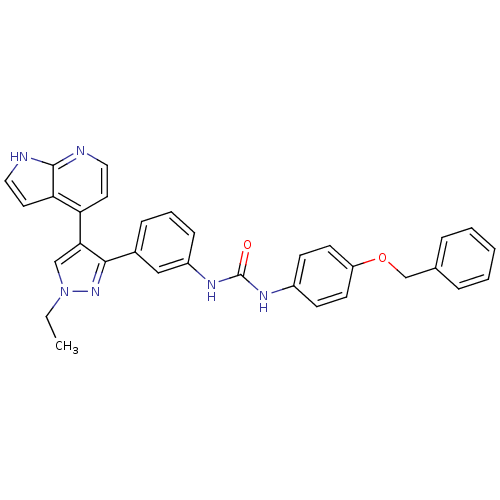
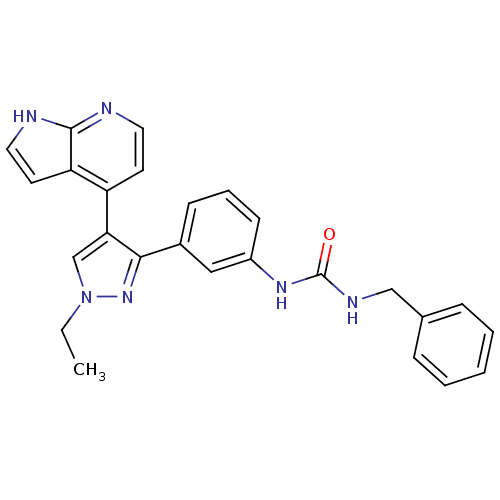
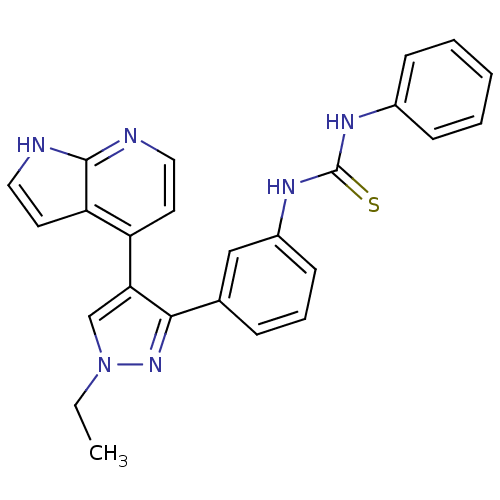
Report error Found 20 Enz. Inhib. hit(s) with all data for entry = 50038648
Affinity DataIC50: 2.10nMAssay Description:Inhibition of B-Raf by fluorescence anisotropy binding assayMore data for this Ligand-Target Pair
Affinity DataIC50: 2.5nMAssay Description:Inhibition of B-Raf by fluorescence anisotropy binding assayMore data for this Ligand-Target Pair
Affinity DataIC50: 3.90nMAssay Description:Inhibition of B-Raf by fluorescence anisotropy binding assayMore data for this Ligand-Target Pair
Affinity DataIC50: 5.30nMAssay Description:Inhibition of B-Raf by fluorescence anisotropy binding assayMore data for this Ligand-Target Pair
Affinity DataIC50: 5.40nMAssay Description:Inhibition of B-Raf by fluorescence anisotropy binding assayMore data for this Ligand-Target Pair
Affinity DataIC50: 10.5nMAssay Description:Inhibition of B-Raf by fluorescence anisotropy binding assayMore data for this Ligand-Target Pair
Affinity DataIC50: 17.4nMAssay Description:Inhibition of B-Raf by fluorescence anisotropy binding assayMore data for this Ligand-Target Pair
Affinity DataIC50: 25.7nMAssay Description:Inhibition of B-Raf by fluorescence anisotropy binding assayMore data for this Ligand-Target Pair
Affinity DataIC50: 33.9nMAssay Description:Inhibition of B-Raf by fluorescence anisotropy binding assayMore data for this Ligand-Target Pair
Affinity DataIC50: 41.7nMAssay Description:Inhibition of B-Raf by fluorescence anisotropy binding assayMore data for this Ligand-Target Pair
Affinity DataIC50: 45.7nMAssay Description:Inhibition of B-Raf by fluorescence anisotropy binding assayMore data for this Ligand-Target Pair
Affinity DataIC50: 63nMAssay Description:Inhibition of B-Raf-mediated phosphorylation of MEK1 in mouse 3T3 cells after 2 hrsMore data for this Ligand-Target Pair
Affinity DataIC50: 79.4nMAssay Description:Inhibition of B-Raf by fluorescence anisotropy binding assayMore data for this Ligand-Target Pair
Affinity DataIC50: 110nMAssay Description:Inhibition of B-Raf by fluorescence anisotropy binding assayMore data for this Ligand-Target Pair
Affinity DataIC50: 170nMAssay Description:Inhibition of B-Raf by fluorescence anisotropy binding assayMore data for this Ligand-Target Pair
Affinity DataIC50: 331nMAssay Description:Inhibition of B-Raf by fluorescence anisotropy binding assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.26E+3nMAssay Description:Inhibition of CYP2C9More data for this Ligand-Target Pair
Affinity DataIC50: 5.01E+3nMAssay Description:Inhibition of CYP2D6More data for this Ligand-Target Pair