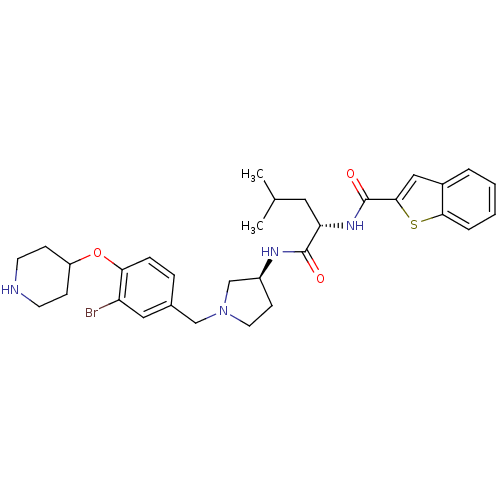
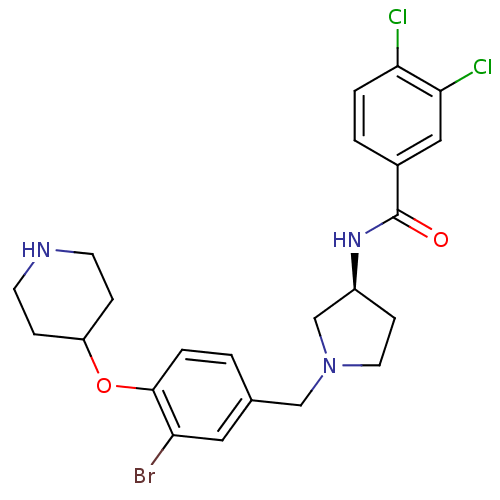
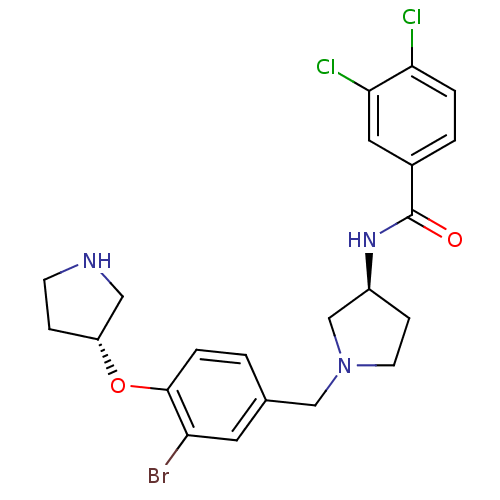
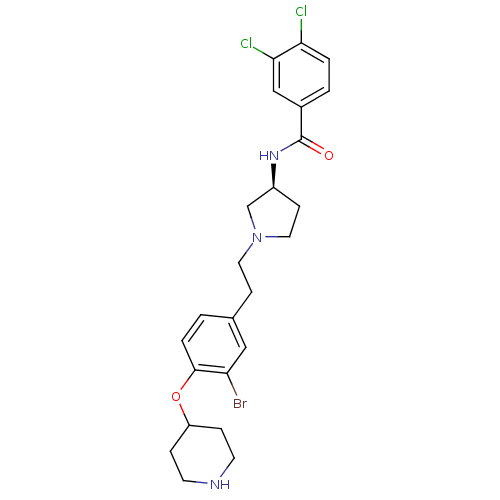
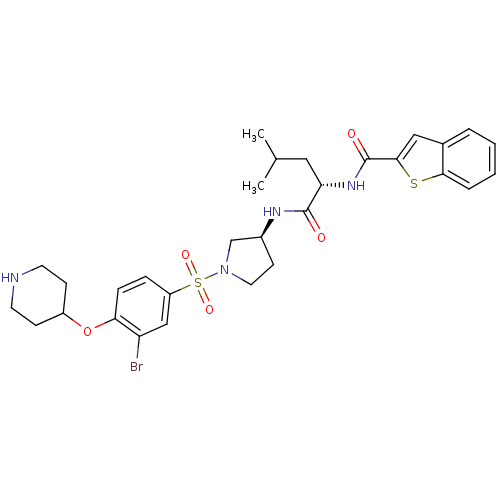
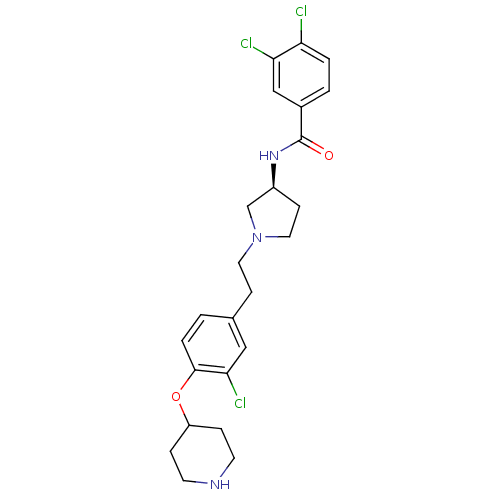
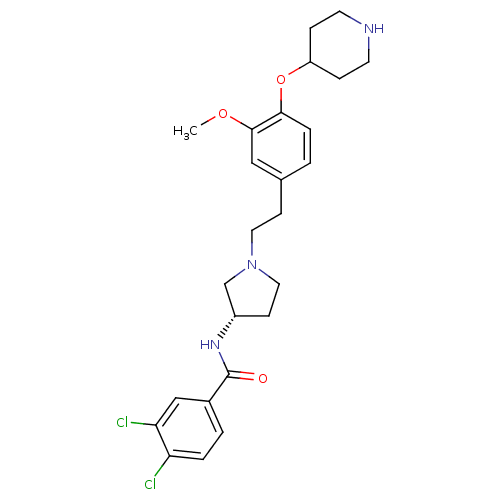
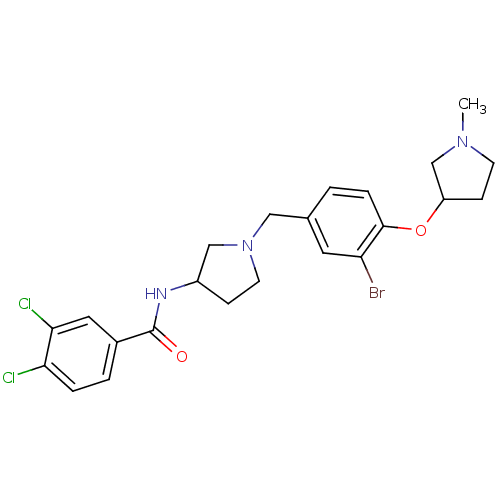
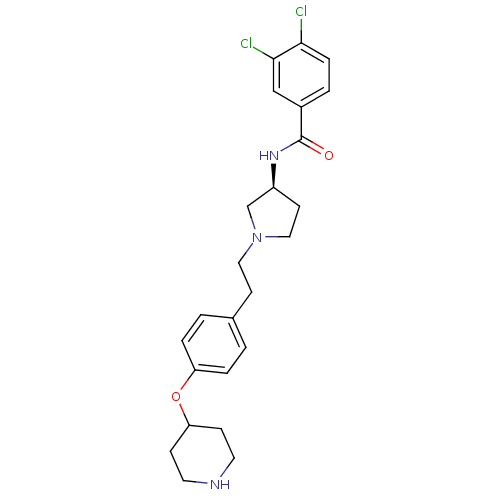
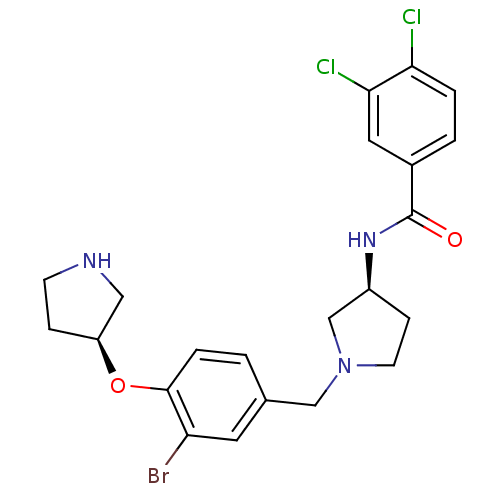
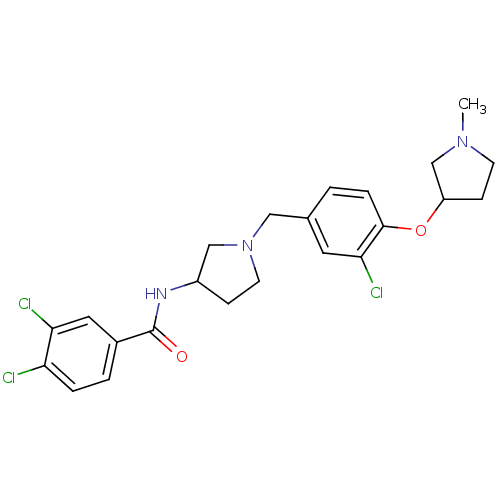
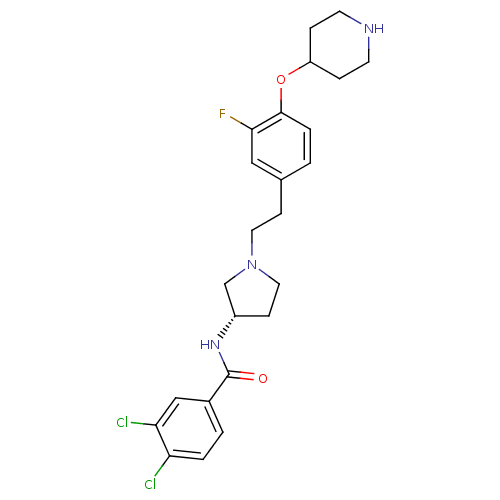
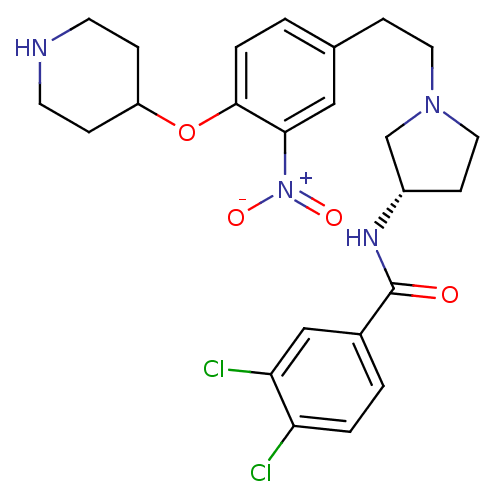
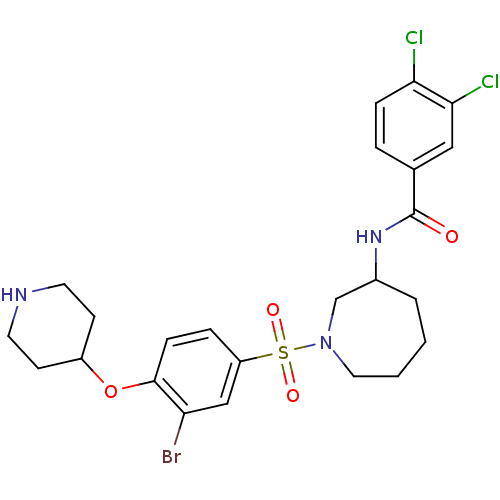
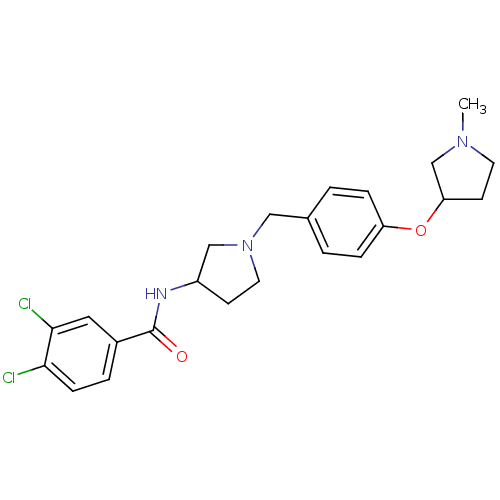
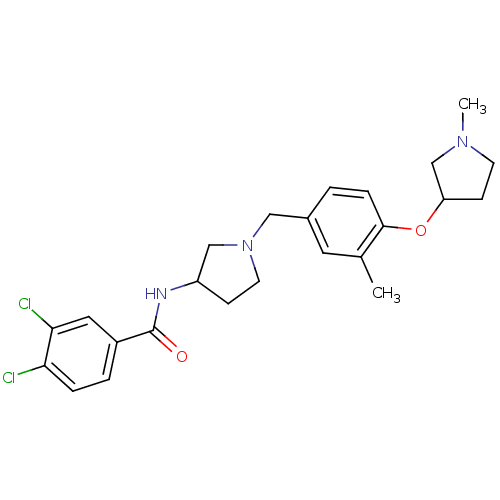
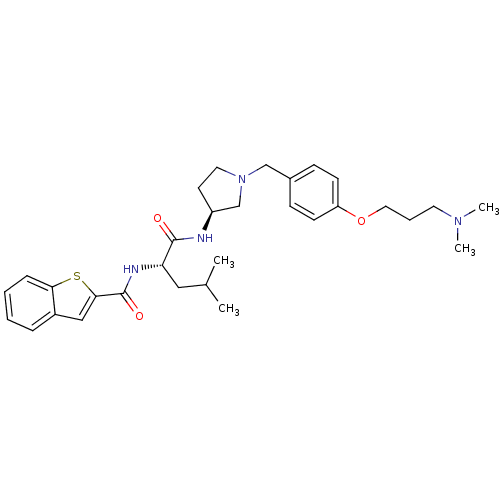
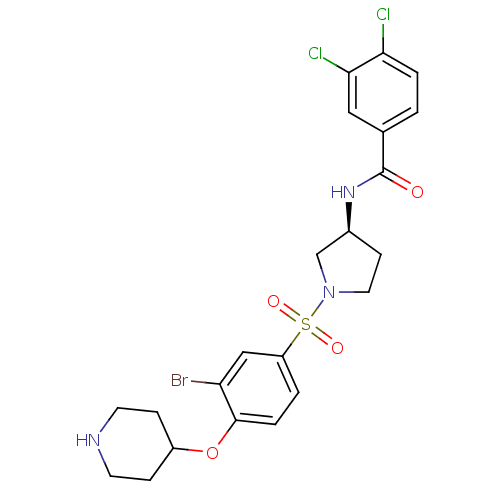
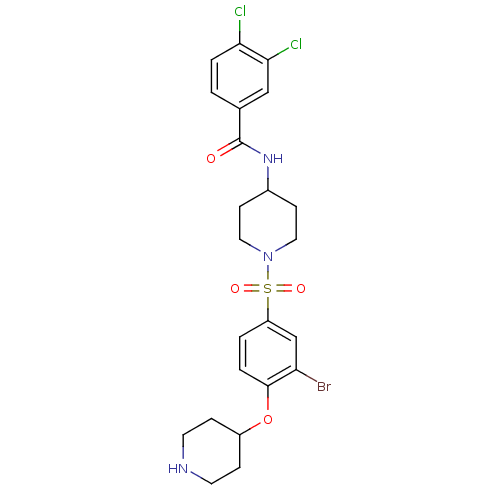
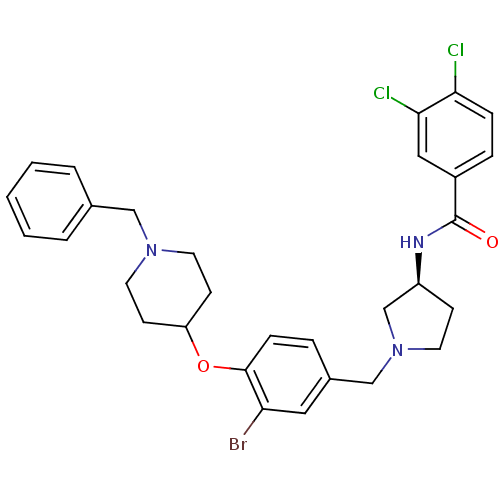
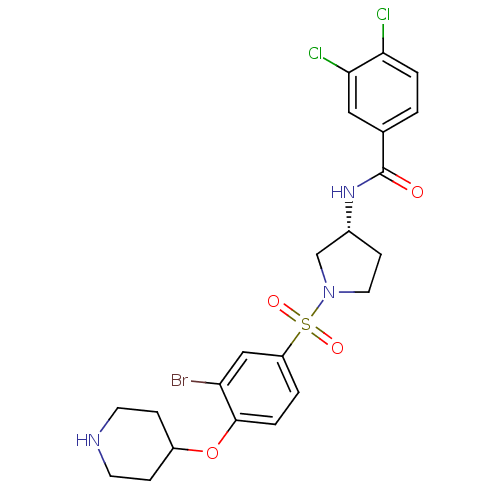
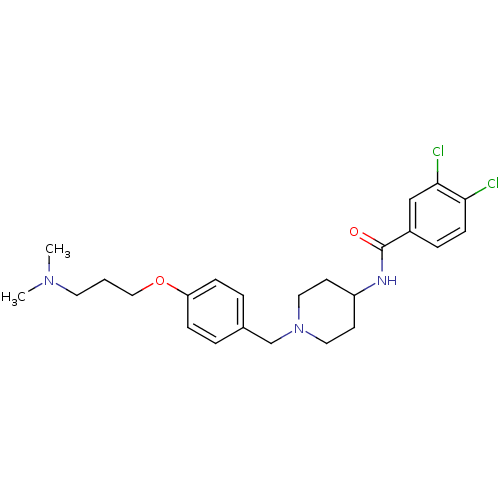
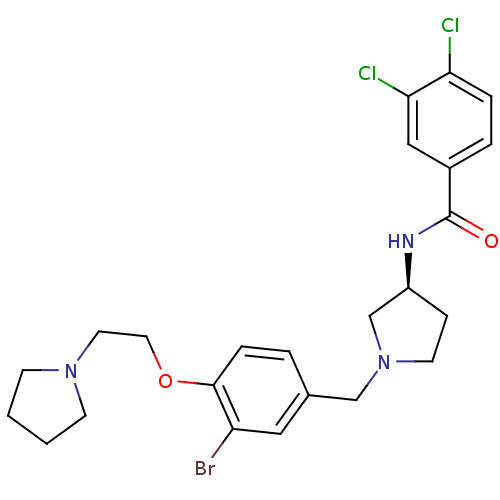
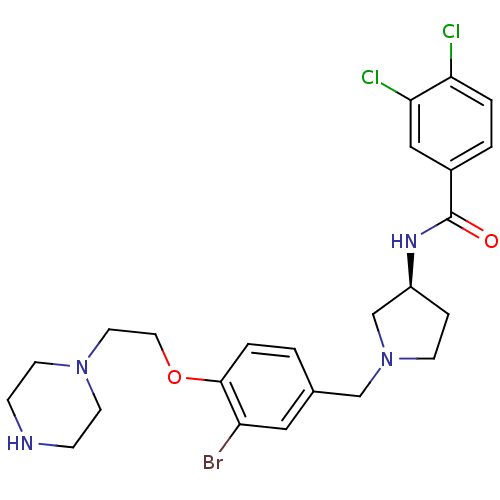
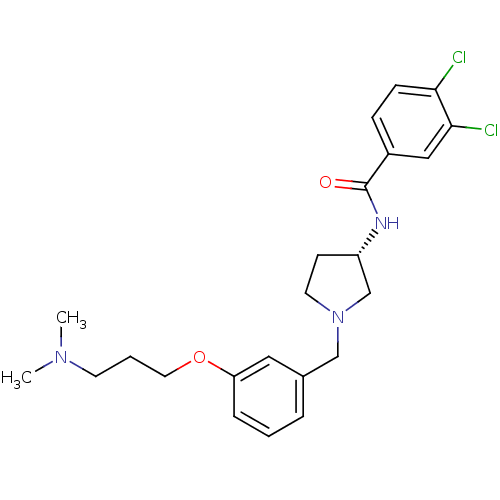
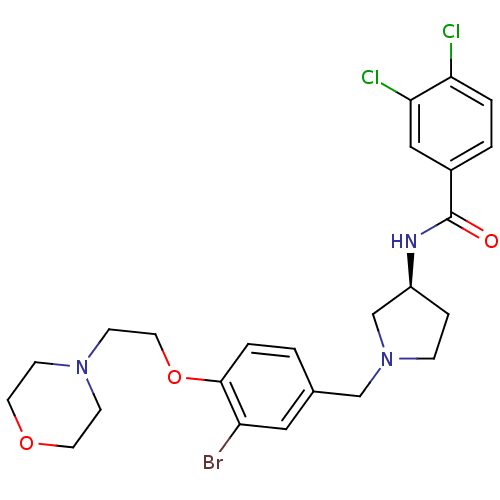
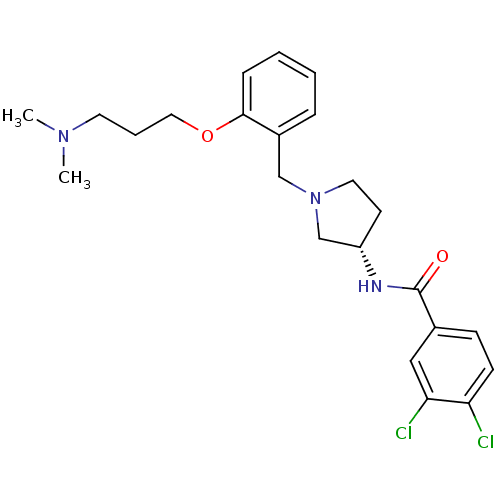
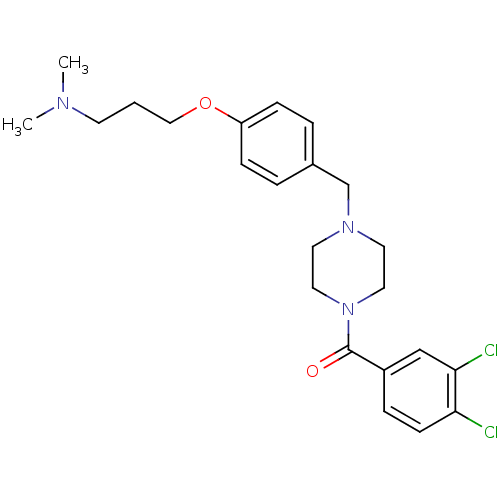
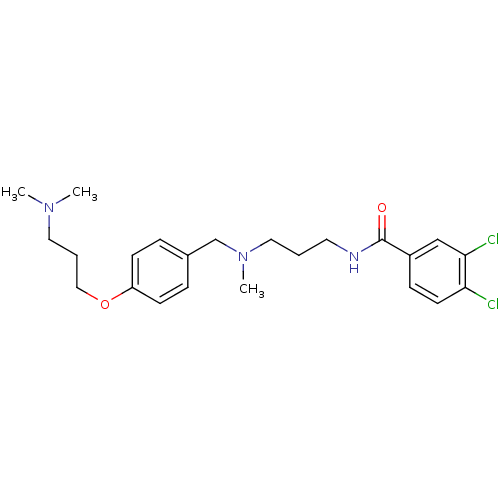
Report error Found 47 Enz. Inhib. hit(s) with all data for entry = 50026465
Affinity DataKi: 3nMAssay Description:Antagonist activity at human recombinant urotensin 2 receptor expressed in HEK293 cells assessed as inhibition of urotensin 2-induced calcium mobiliz...More data for this Ligand-Target Pair
Affinity DataKi: 4nMAssay Description:Displacement of [I125]hU2 from human recombinant urotensin 2 receptor expressed in HEK293 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 4nMAssay Description:Antagonist activity at human recombinant urotensin 2 receptor expressed in HEK293 cells assessed as inhibition of urotensin 2-induced calcium mobiliz...More data for this Ligand-Target Pair
Affinity DataKi: 5.5nMAssay Description:Antagonist activity at human recombinant urotensin 2 receptor expressed in HEK293 cells assessed as inhibition of urotensin 2-induced calcium mobiliz...More data for this Ligand-Target Pair
Affinity DataKi: 10nMAssay Description:Antagonist activity at human recombinant urotensin 2 receptor expressed in HEK293 cells assessed as inhibition of urotensin 2-induced calcium mobiliz...More data for this Ligand-Target Pair
Affinity DataKi: 12nMAssay Description:Antagonist activity at human recombinant urotensin 2 receptor expressed in HEK293 cells assessed as inhibition of urotensin 2-induced calcium mobiliz...More data for this Ligand-Target Pair
Affinity DataKi: 15nMAssay Description:Antagonist activity at human recombinant urotensin 2 receptor expressed in HEK293 cells assessed as inhibition of urotensin 2-induced calcium mobiliz...More data for this Ligand-Target Pair
Affinity DataKi: 18nMAssay Description:Antagonist activity at human recombinant urotensin 2 receptor expressed in HEK293 cells assessed as inhibition of urotensin 2-induced calcium mobiliz...More data for this Ligand-Target Pair
Affinity DataKi: 36nMAssay Description:Antagonist activity at human recombinant urotensin 2 receptor expressed in HEK293 cells assessed as inhibition of urotensin 2-induced calcium mobiliz...More data for this Ligand-Target Pair
Affinity DataKi: 38nMAssay Description:Binding affinity to 5HT1A receptorMore data for this Ligand-Target Pair
Affinity DataKi: 41nMAssay Description:Antagonist activity at human recombinant urotensin 2 receptor expressed in HEK293 cells assessed as inhibition of urotensin 2-induced calcium mobiliz...More data for this Ligand-Target Pair
Affinity DataKi: 42nMAssay Description:Antagonist activity at human recombinant urotensin 2 receptor expressed in HEK293 cells assessed as inhibition of urotensin 2-induced calcium mobiliz...More data for this Ligand-Target Pair
Affinity DataKi: 44nMAssay Description:Antagonist activity at human recombinant urotensin 2 receptor expressed in HEK293 cells assessed as inhibition of urotensin 2-induced calcium mobiliz...More data for this Ligand-Target Pair
Affinity DataKi: 47nMAssay Description:Antagonist activity at human recombinant urotensin 2 receptor expressed in HEK293 cells assessed as inhibition of urotensin 2-induced calcium mobiliz...More data for this Ligand-Target Pair
Affinity DataKi: 61nMAssay Description:Antagonist activity at human recombinant urotensin 2 receptor expressed in HEK293 cells assessed as inhibition of urotensin 2-induced calcium mobiliz...More data for this Ligand-Target Pair
Affinity DataKi: 62nMAssay Description:Antagonist activity at human recombinant urotensin 2 receptor expressed in HEK293 cells assessed as inhibition of urotensin 2-induced calcium mobiliz...More data for this Ligand-Target Pair
Affinity DataKi: 69nMAssay Description:Antagonist activity at human recombinant urotensin 2 receptor expressed in HEK293 cells assessed as inhibition of urotensin 2-induced calcium mobiliz...More data for this Ligand-Target Pair
Affinity DataKi: 71nMAssay Description:Antagonist activity at human recombinant urotensin 2 receptor expressed in HEK293 cells assessed as inhibition of urotensin 2-induced calcium mobiliz...More data for this Ligand-Target Pair
Affinity DataKi: 85nMAssay Description:Binding affinity at 5HT2C receptorMore data for this Ligand-Target Pair
Affinity DataKi: 97nMAssay Description:Antagonist activity at human recombinant urotensin 2 receptor expressed in HEK293 cells assessed as inhibition of urotensin 2-induced calcium mobiliz...More data for this Ligand-Target Pair
Affinity DataKi: 100nMAssay Description:Binding affinity to dopamine D2 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 108nMAssay Description:Antagonist activity at human recombinant urotensin 2 receptor expressed in HEK293 cells assessed as inhibition of urotensin 2-induced calcium mobiliz...More data for this Ligand-Target Pair
Affinity DataKi: 110nMAssay Description:Antagonist activity at human recombinant urotensin 2 receptor expressed in HEK293 cells assessed as inhibition of urotensin 2-induced calcium mobiliz...More data for this Ligand-Target Pair
Affinity DataKi: 120nMAssay Description:Antagonist activity at human recombinant urotensin 2 receptor expressed in HEK293 cells assessed as inhibition of urotensin 2-induced calcium mobiliz...More data for this Ligand-Target Pair
Affinity DataKi: 140nMAssay Description:Antagonist activity at human recombinant urotensin 2 receptor expressed in HEK293 cells assessed as inhibition of urotensin 2-induced calcium mobiliz...More data for this Ligand-Target Pair
Affinity DataKi: 140nMAssay Description:Binding affinity to 5HT1D receptorMore data for this Ligand-Target Pair
Affinity DataKi: 140nMAssay Description:Binding affinity at 5HT2B receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 170nMAssay Description:Antagonist activity at human recombinant urotensin 2 receptor expressed in HEK293 cells assessed as inhibition of urotensin 2-induced calcium mobiliz...More data for this Ligand-Target Pair
Affinity DataKi: 200nMAssay Description:Antagonist activity at human recombinant urotensin 2 receptor expressed in HEK293 cells assessed as inhibition of urotensin 2-induced calcium mobiliz...More data for this Ligand-Target Pair
Affinity DataKi: 230nMAssay Description:Antagonist activity at human recombinant urotensin 2 receptor expressed in HEK293 cells assessed as inhibition of urotensin 2-induced calcium mobiliz...More data for this Ligand-Target Pair
Affinity DataKi: 320nMAssay Description:Antagonist activity at human recombinant urotensin 2 receptor expressed in HEK293 cells assessed as inhibition of urotensin 2-induced calcium mobiliz...More data for this Ligand-Target Pair
Affinity DataKi: 350nMAssay Description:Antagonist activity at human recombinant urotensin 2 receptor expressed in HEK293 cells assessed as inhibition of urotensin 2-induced calcium mobiliz...More data for this Ligand-Target Pair
Affinity DataKi: 380nMAssay Description:Antagonist activity at human recombinant urotensin 2 receptor expressed in HEK293 cells assessed as inhibition of urotensin 2-induced calcium mobiliz...More data for this Ligand-Target Pair
Affinity DataKi: 500nMAssay Description:Antagonist activity at human recombinant urotensin 2 receptor expressed in HEK293 cells assessed as inhibition of urotensin 2-induced calcium mobiliz...More data for this Ligand-Target Pair
Affinity DataKi: 1.00E+3nMAssay Description:Antagonist activity at human recombinant urotensin 2 receptor expressed in HEK293 cells assessed as inhibition of urotensin 2-induced calcium mobiliz...More data for this Ligand-Target Pair
Affinity DataKi: 1.50E+3nMAssay Description:Antagonist activity at human recombinant urotensin 2 receptor expressed in HEK293 cells assessed as inhibition of urotensin 2-induced calcium mobiliz...More data for this Ligand-Target Pair
Affinity DataIC50: 1.90E+3nMAssay Description:Inhibition of CYP3A4More data for this Ligand-Target Pair
Affinity DataIC50: 2.20E+3nMAssay Description:Inhibition of CYP2D6More data for this Ligand-Target Pair
Affinity DataKi: >3.00E+3nMAssay Description:Antagonist activity at human recombinant urotensin 2 receptor expressed in HEK293 cells assessed as inhibition of urotensin 2-induced calcium mobiliz...More data for this Ligand-Target Pair
Affinity DataKi: 3.50E+3nMAssay Description:Antagonist activity at human recombinant urotensin 2 receptor expressed in HEK293 cells assessed as inhibition of urotensin 2-induced calcium mobiliz...More data for this Ligand-Target Pair
Affinity DataKi: 4.10E+3nMAssay Description:Binding affinity to 5HT6 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 7.20E+3nMAssay Description:Binding affinity to 5HT1F receptorMore data for this Ligand-Target Pair
Affinity DataKi: >1.00E+4nMAssay Description:Binding affinity to 5HT1E receptorMore data for this Ligand-Target Pair
Affinity DataKi: >1.00E+4nMAssay Description:Antagonist activity at human recombinant urotensin 2 receptor expressed in HEK293 cells assessed as inhibition of urotensin 2-induced calcium mobiliz...More data for this Ligand-Target Pair
Affinity DataKi: >1.00E+4nMAssay Description:Antagonist activity at human recombinant urotensin 2 receptor expressed in HEK293 cells assessed as inhibition of urotensin 2-induced calcium mobiliz...More data for this Ligand-Target Pair
Affinity DataKi: >1.00E+4nMAssay Description:Antagonist activity at human recombinant urotensin 2 receptor expressed in HEK293 cells assessed as inhibition of urotensin 2-induced calcium mobiliz...More data for this Ligand-Target Pair
Affinity DataKi: >1.00E+4nMAssay Description:Binding affinity to beta 2 adrenergic receptorMore data for this Ligand-Target Pair