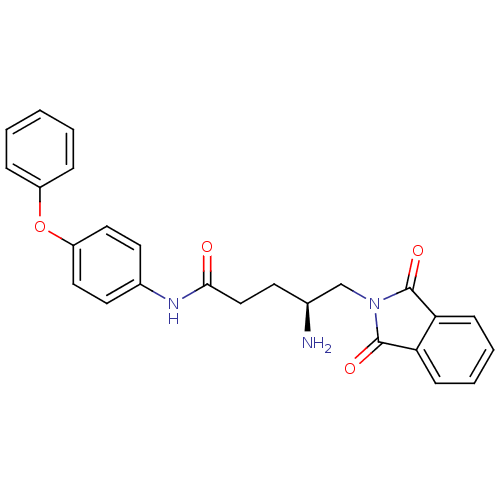
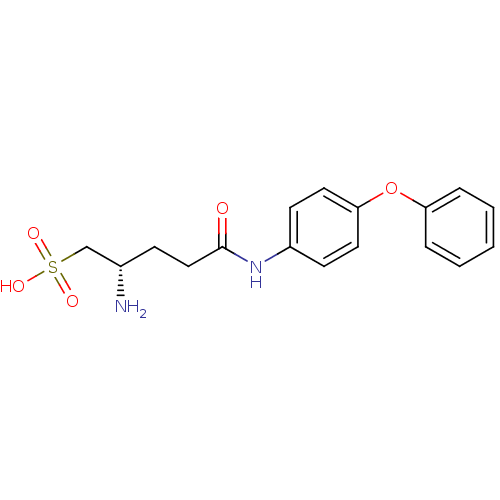
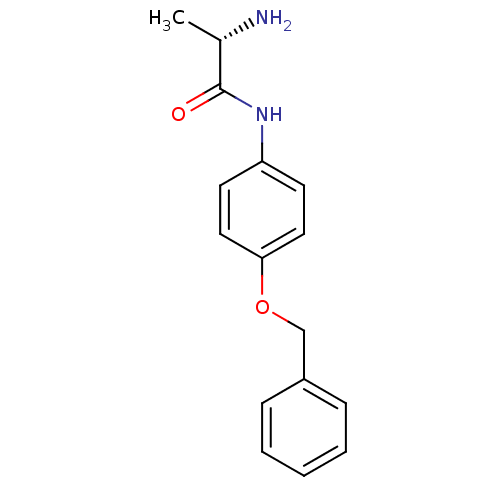
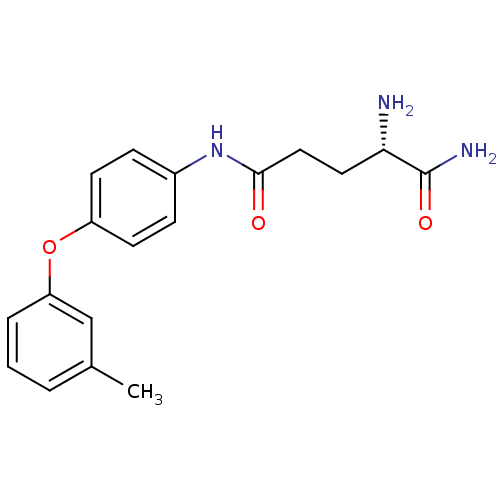
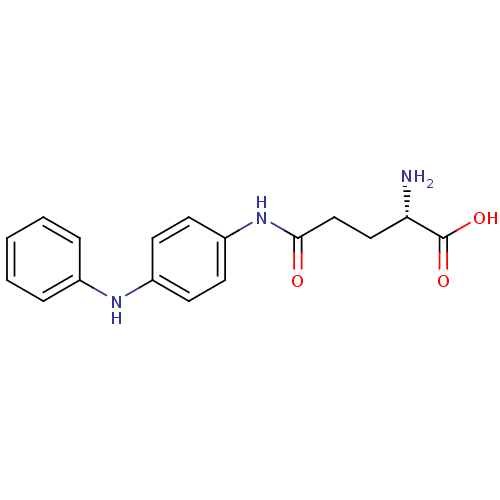
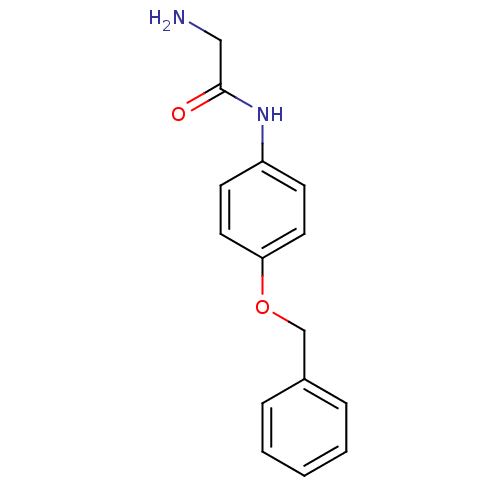
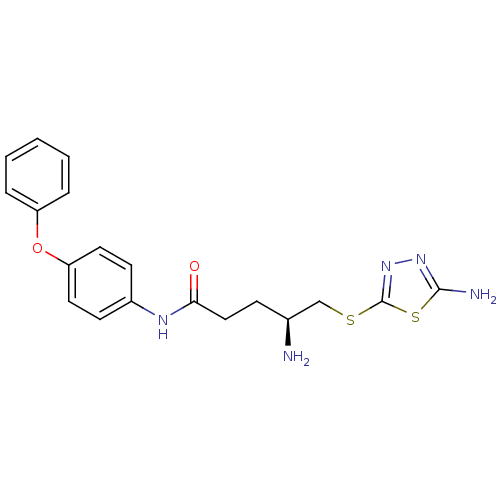
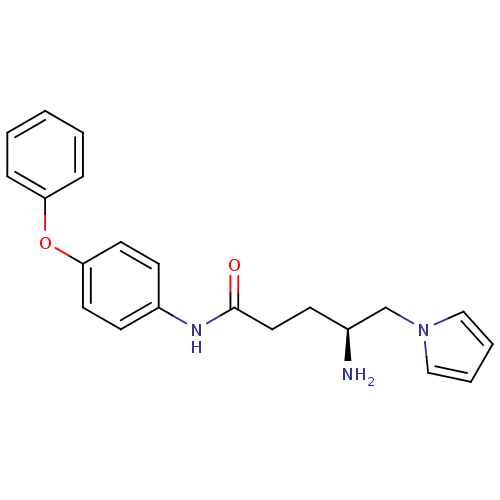
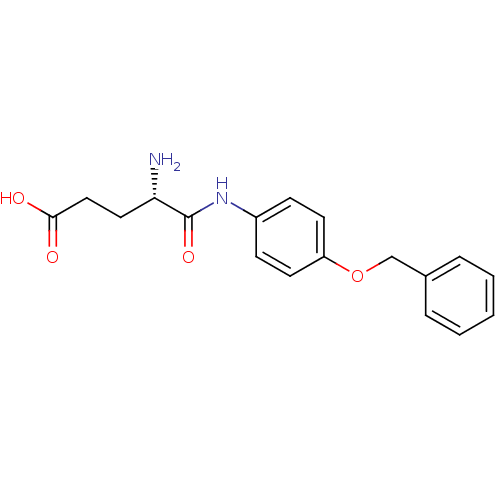
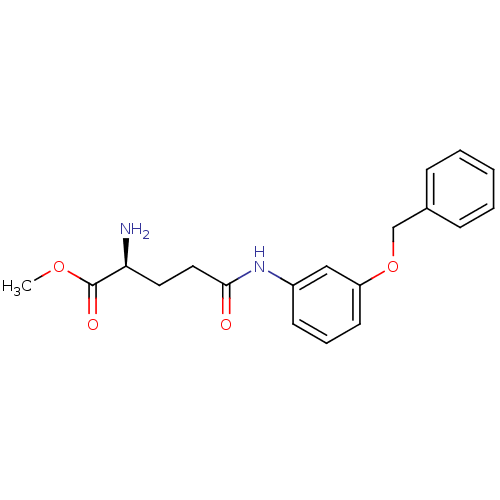
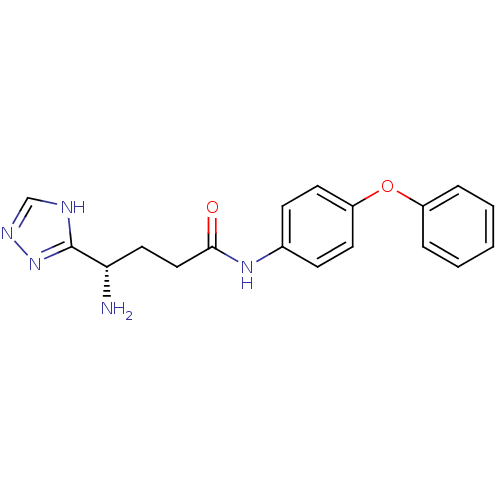
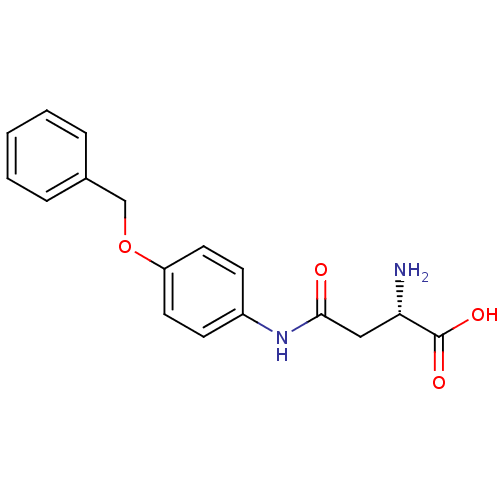
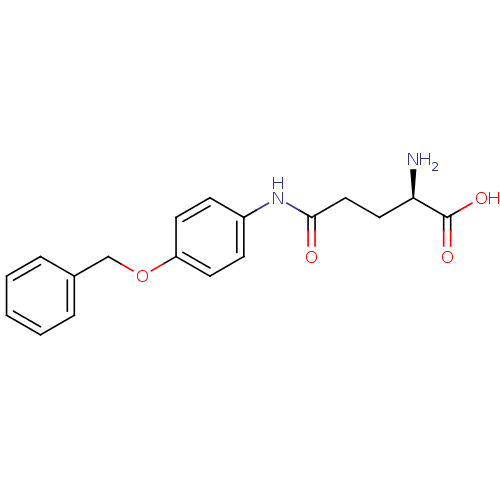
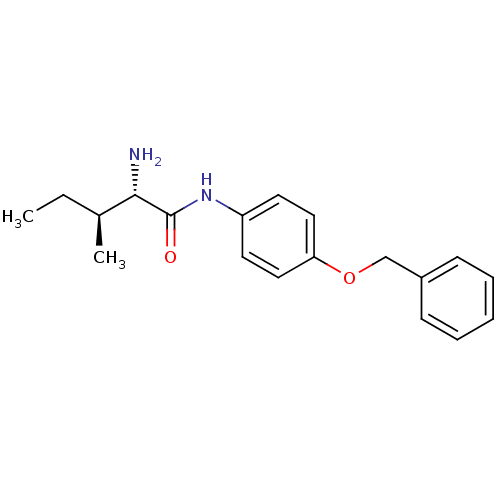
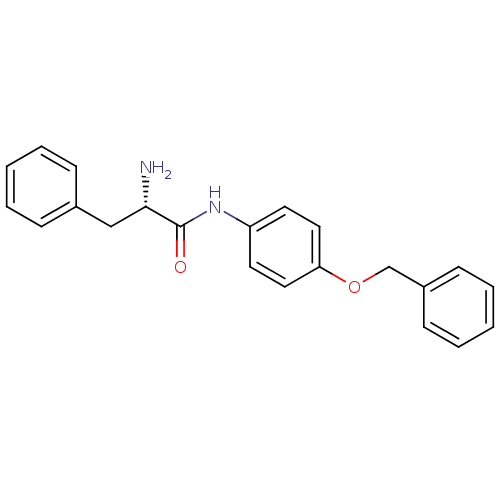
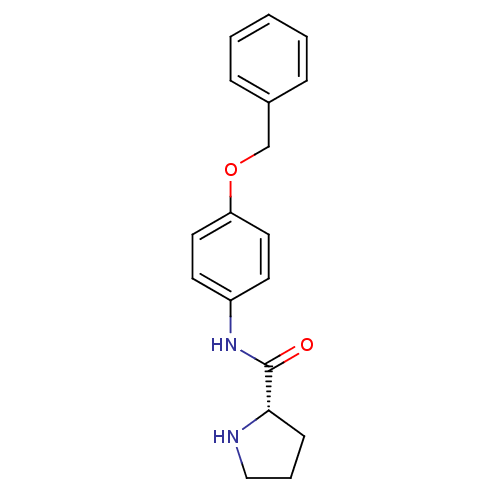
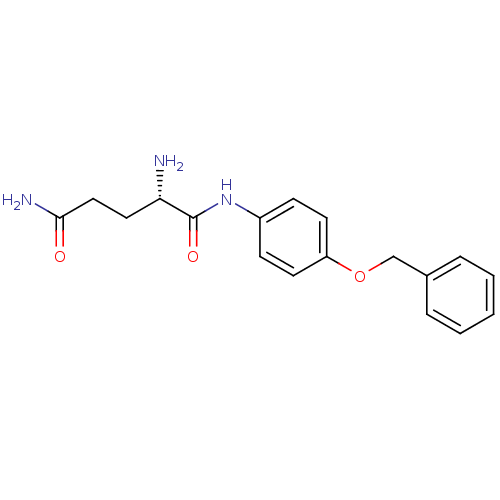
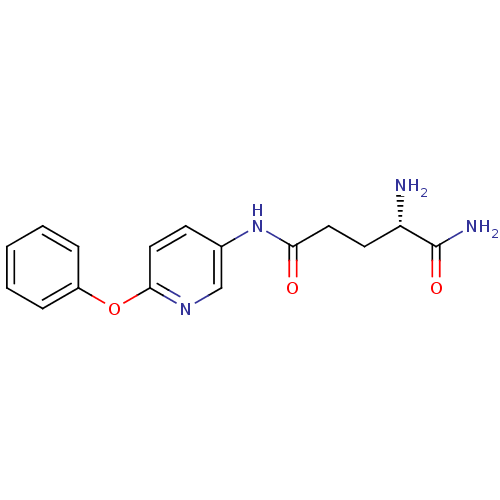
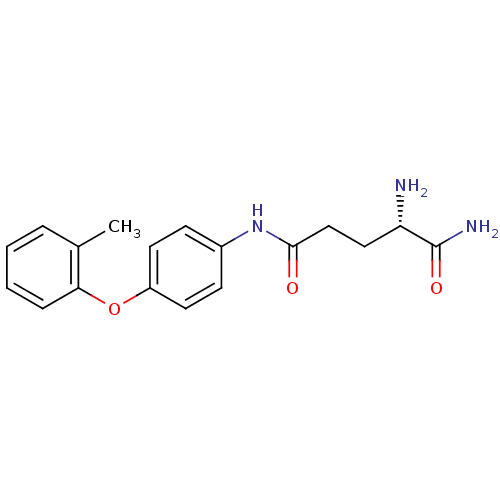
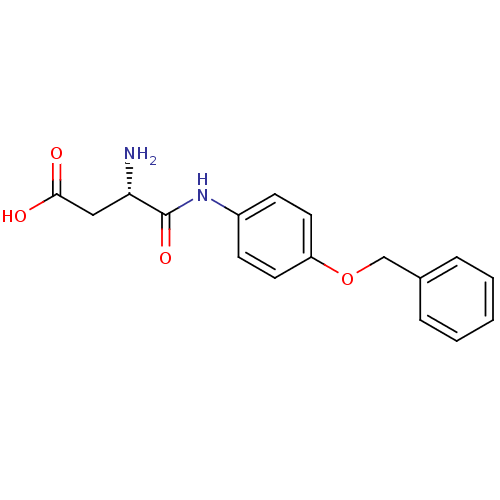
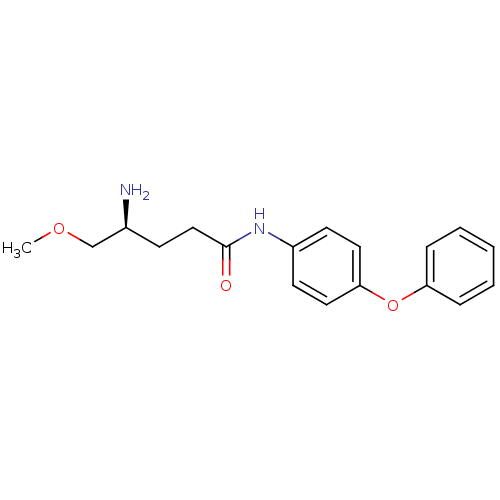
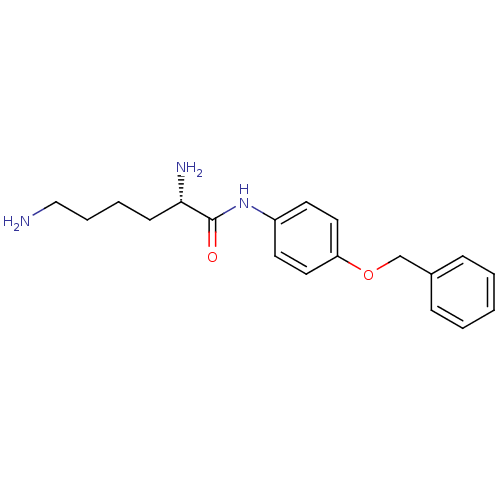
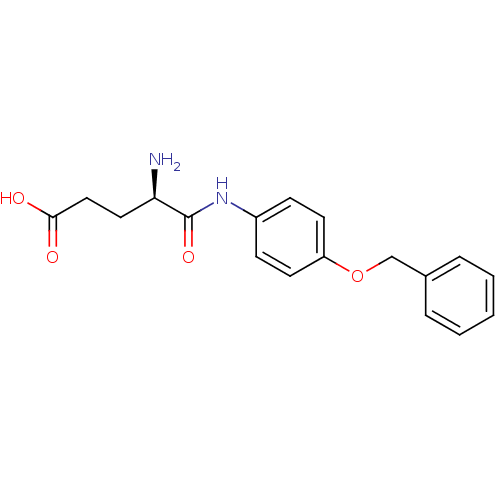
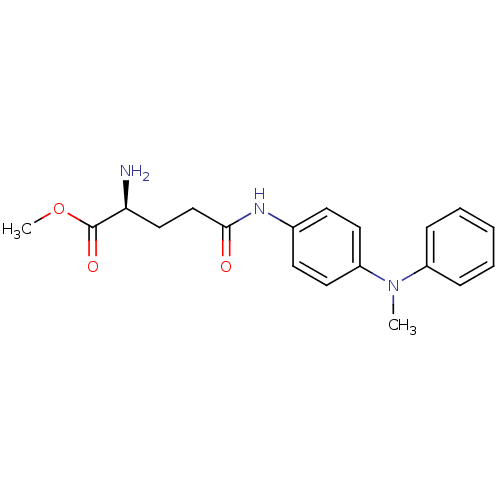
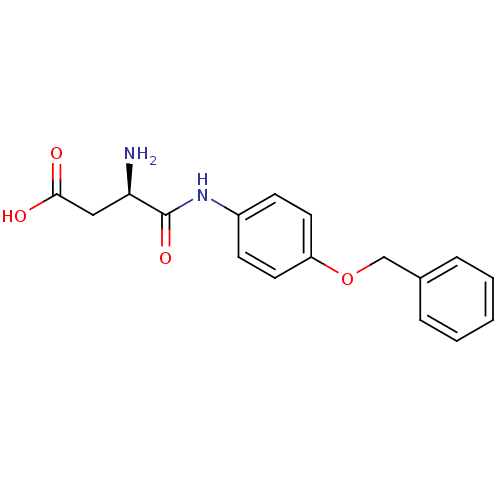
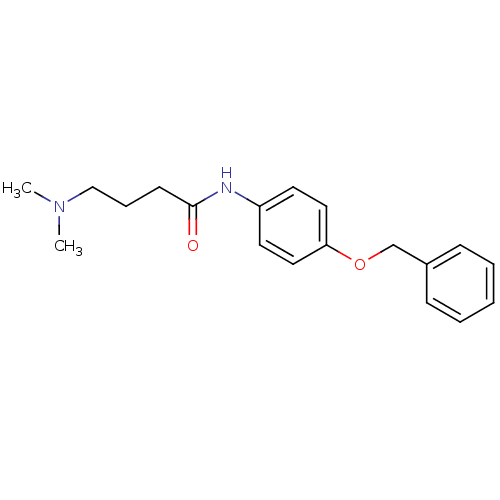
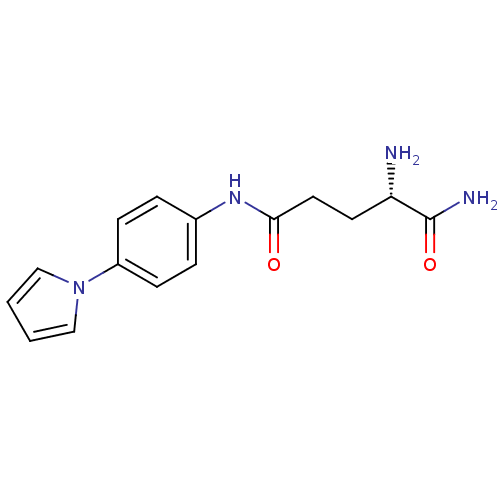
Report error Found 56 Enz. Inhib. hit(s) with all data for entry = 2756
Affinity DataIC50: 6nMAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 13nMAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 14nMAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 17nMAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 17nMAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 18nMAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 19nMpH: 7.5 T: 2°CAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 19nMAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMpH: 7.5 T: 2°CAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 21nMAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 21nMpH: 7.5 T: 2°CAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 22nMAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 22nMAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 22nMAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 23nMpH: 7.5 T: 2°CAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 25nMpH: 7.5 T: 2°CAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 29nMAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 31nMAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 39nMpH: 7.5 T: 2°CAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 46nMAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 60nMpH: 7.5 T: 2°CAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 61nMpH: 7.5 T: 2°CAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 85nMAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 130nMAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMpH: 7.5 T: 2°CAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 210nMpH: 7.5 T: 2°CAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 280nMpH: 7.5 T: 2°CAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 350nMAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 390nMAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 680nMpH: 7.5 T: 2°CAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 730nMpH: 7.5 T: 2°CAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 740nMAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 1.10E+3nMpH: 7.5 T: 2°CAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 1.60E+3nMpH: 7.5 T: 2°CAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 2.00E+3nMpH: 7.5 T: 2°CAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 2.00E+3nMpH: 7.5 T: 2°CAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 2.00E+3nMpH: 7.5 T: 2°CAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 2.00E+3nMpH: 7.5 T: 2°CAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 2.60E+3nMpH: 7.5 T: 2°CAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 2.80E+3nMAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 5.40E+3nMpH: 7.5 T: 2°CAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 5.60E+3nMAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 5.80E+3nMpH: 7.5 T: 2°CAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 7.20E+3nMpH: 7.5 T: 2°CAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 8.00E+3nMAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 9.30E+3nMpH: 7.5 T: 2°CAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 9.40E+3nMpH: 7.5 T: 2°CAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
Affinity DataIC50: 9.70E+3nMpH: 7.5 T: 2°CAssay Description:Compound potency against the peptidase activity of LTA4 hydrolase was measured by inhibition of the hydrolysis of L-alanine-p-nitroanilide to L-alani...More data for this Ligand-Target Pair
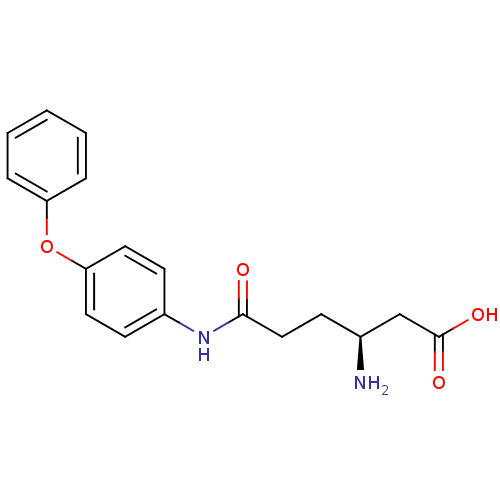
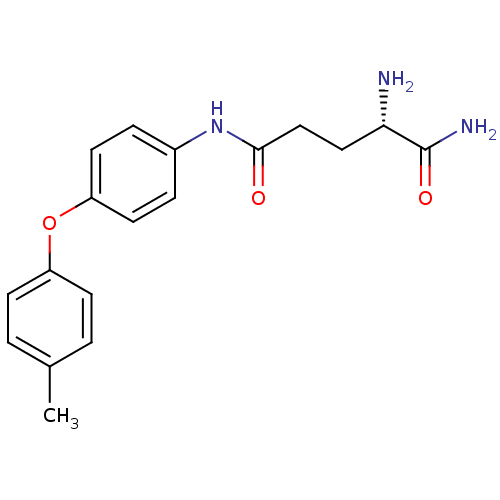
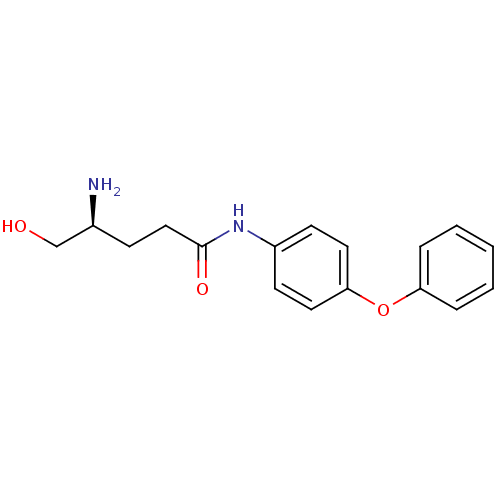
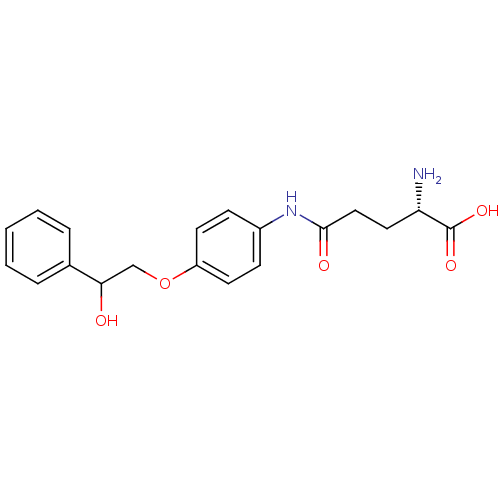
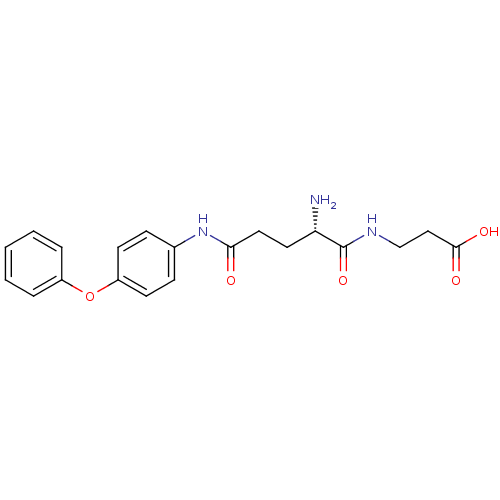
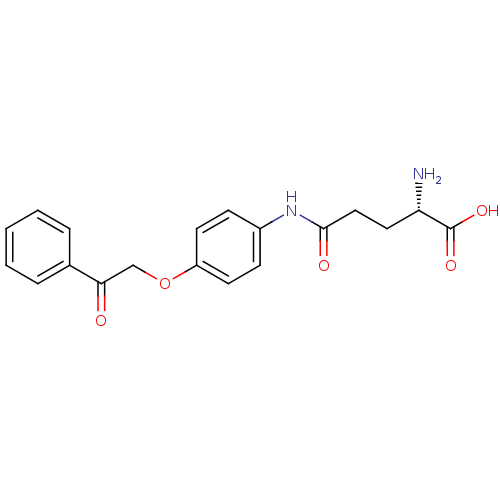
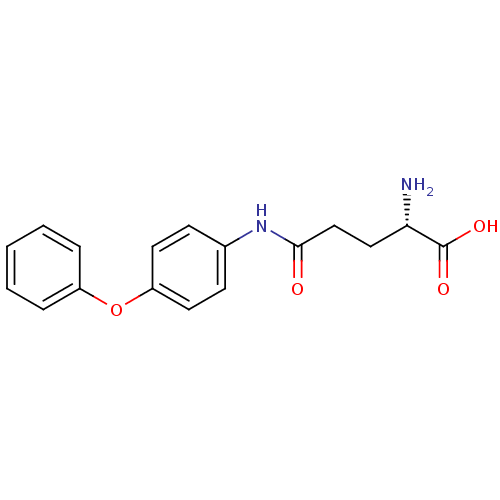
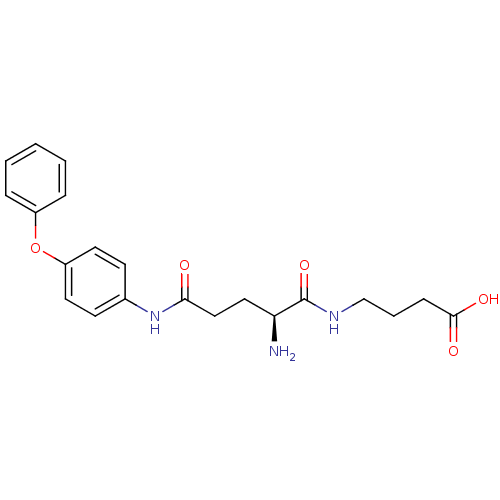
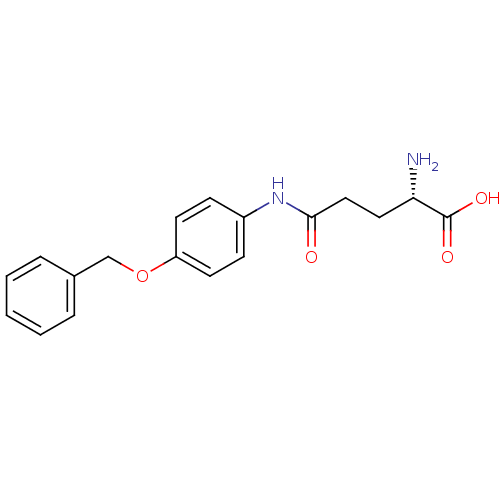
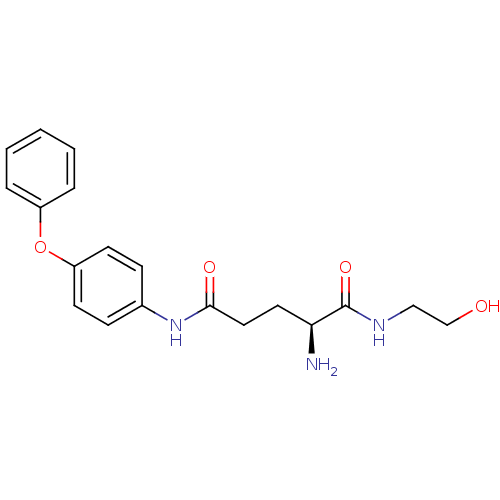
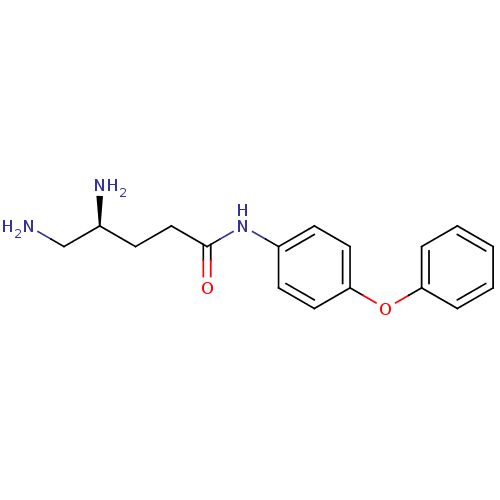
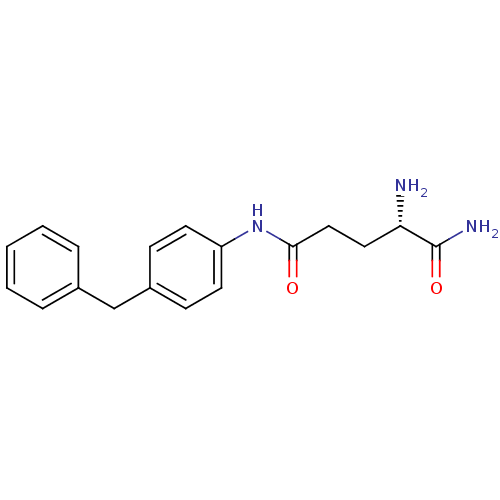
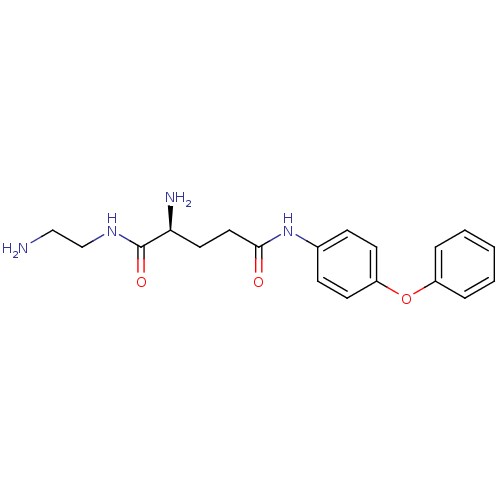
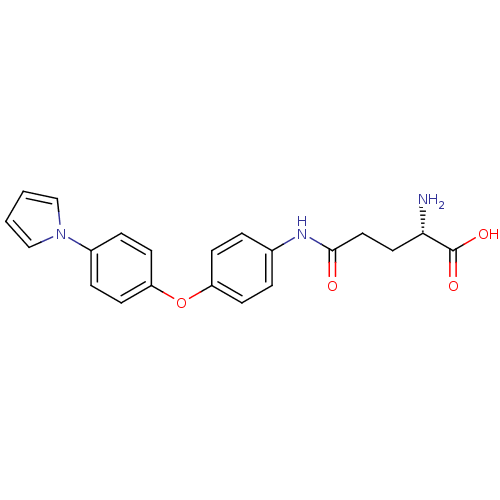
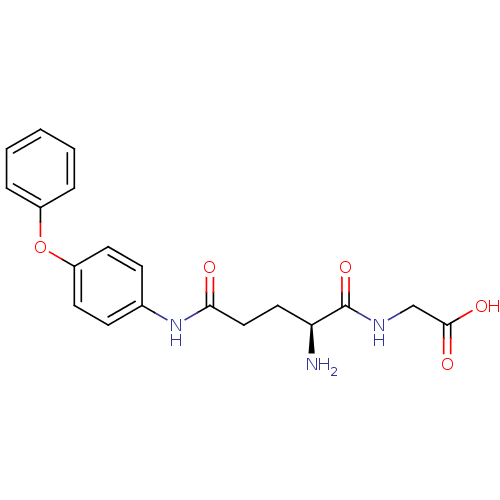
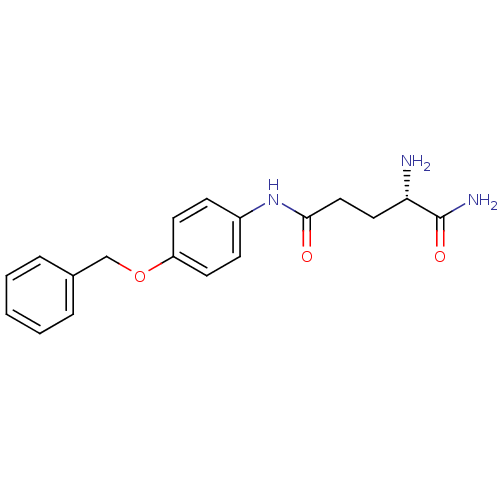
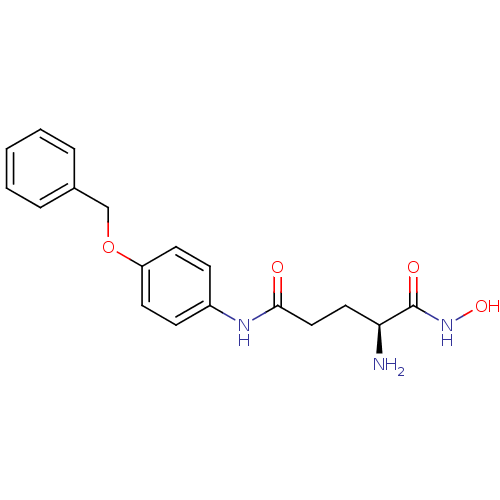
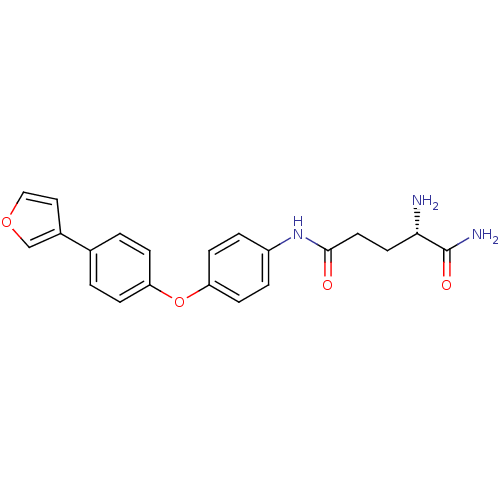
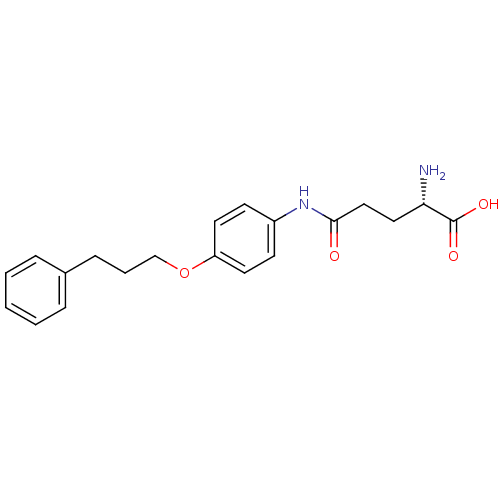
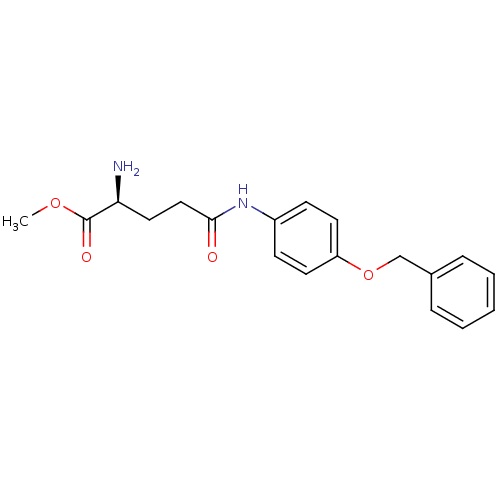
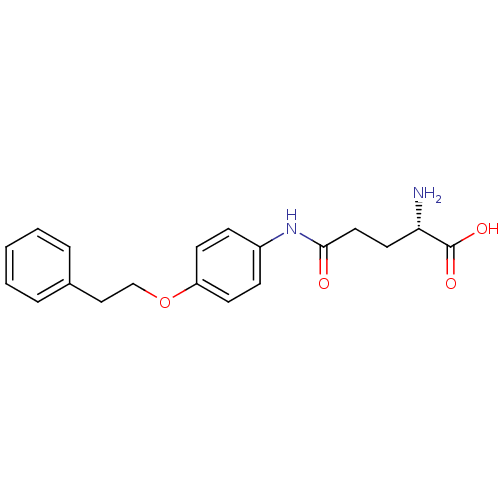
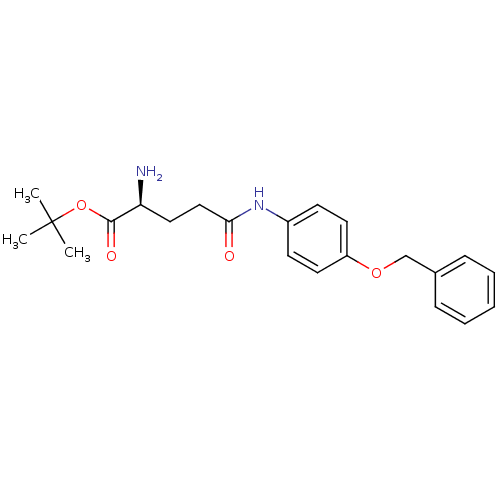
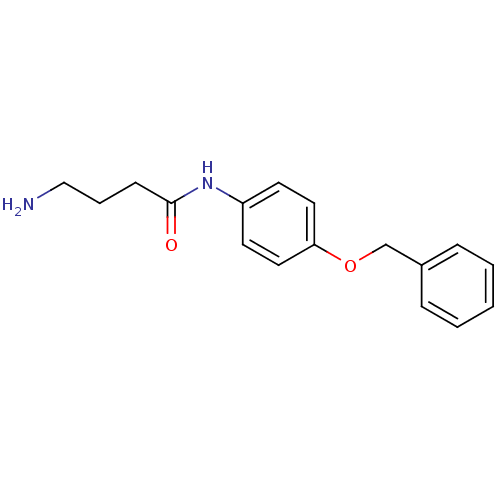
 3D Structure (crystal)
3D Structure (crystal)