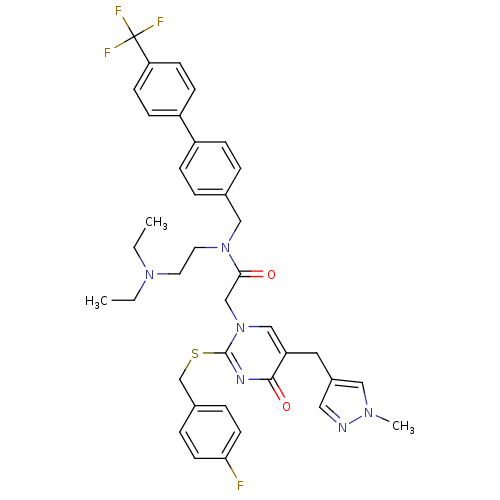
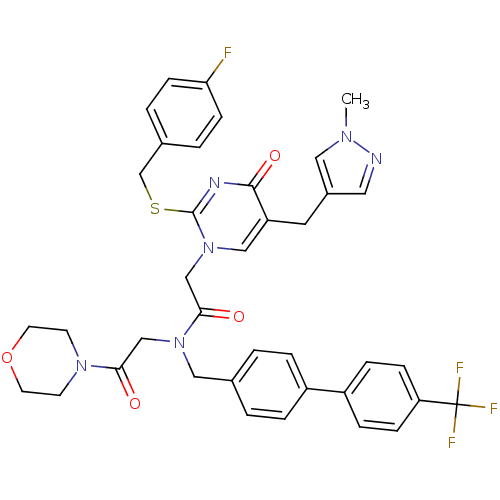
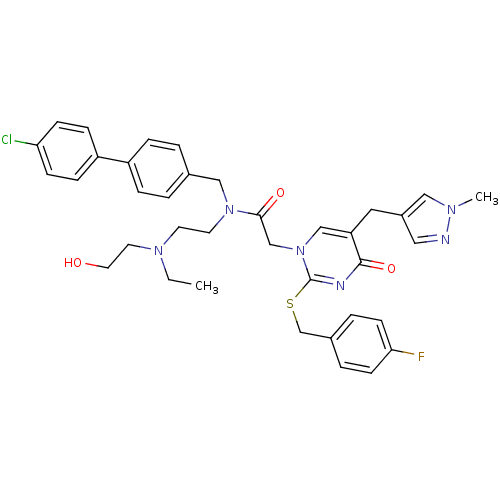
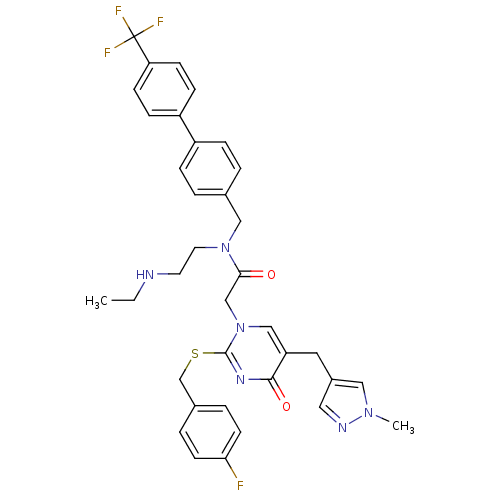
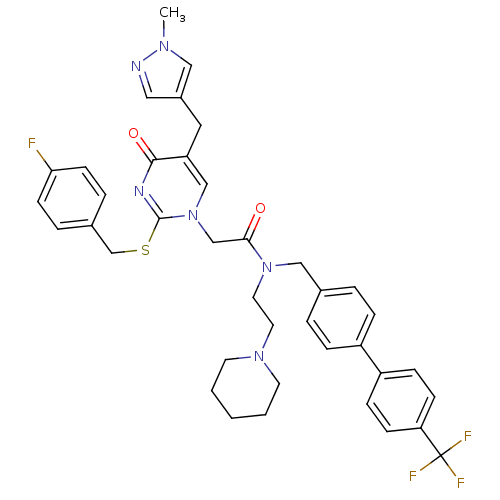
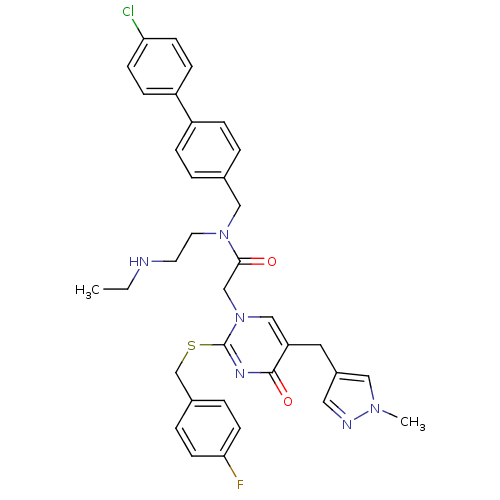
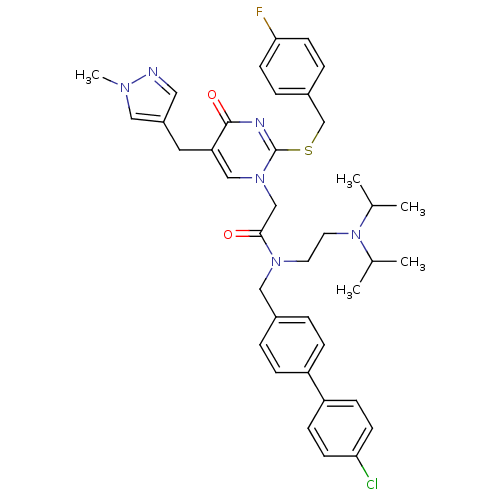
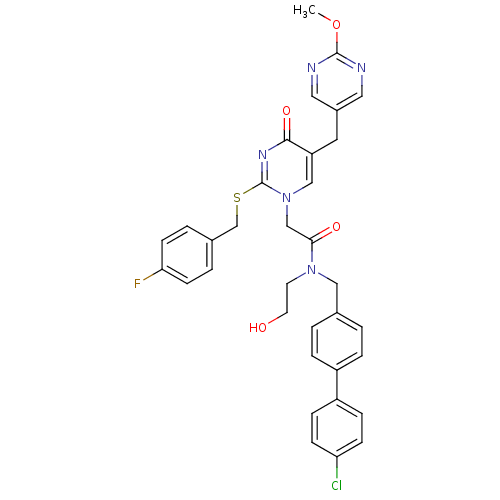
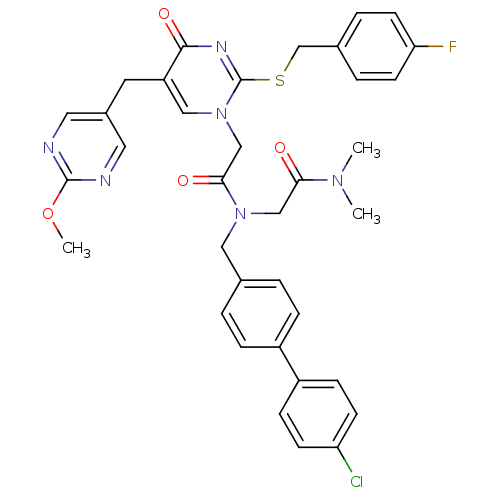
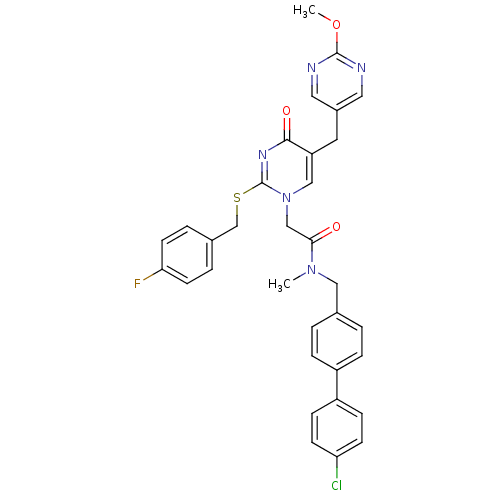
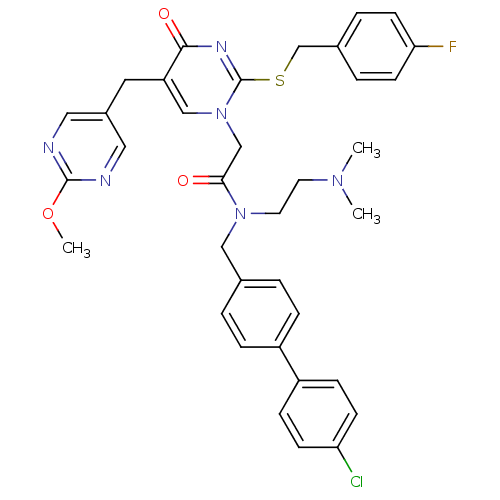
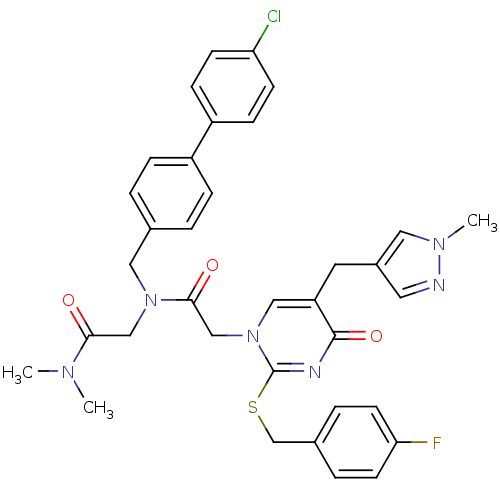
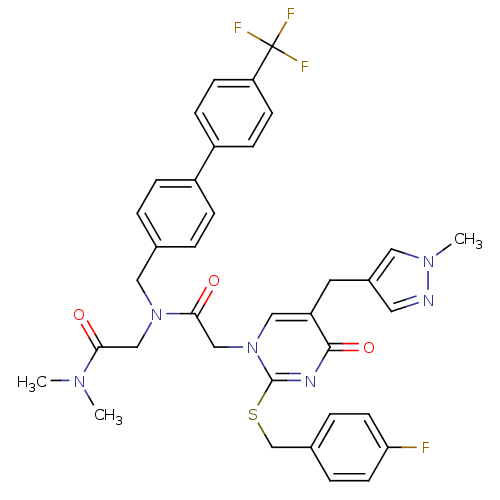
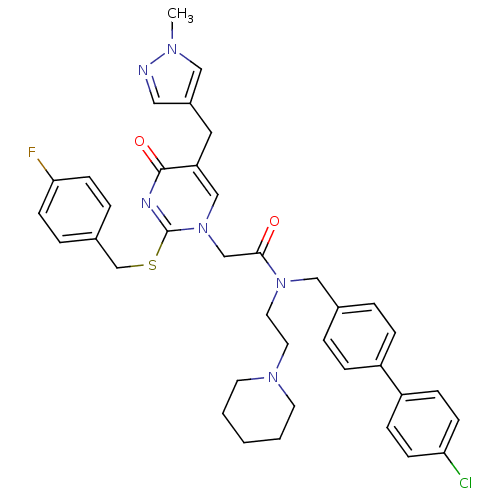
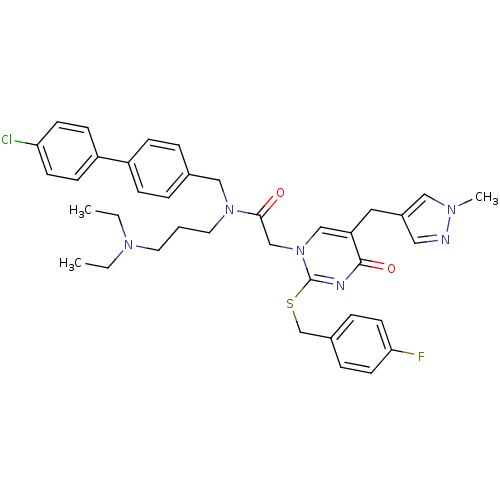
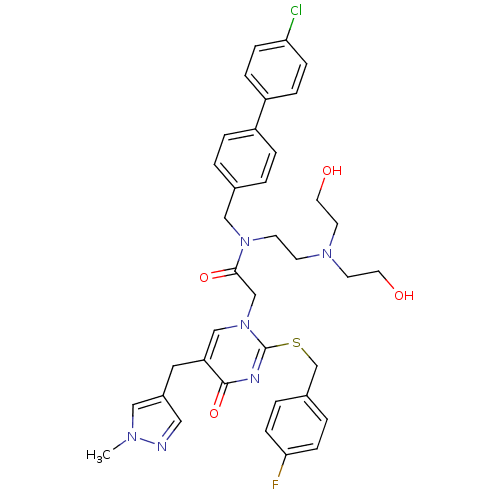
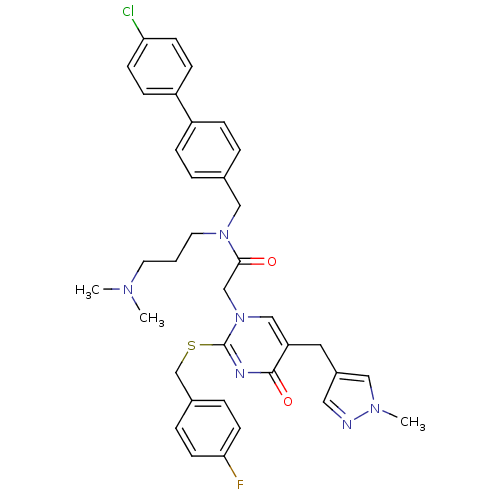
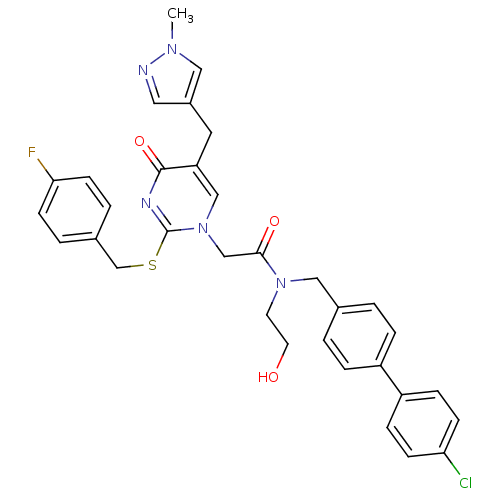
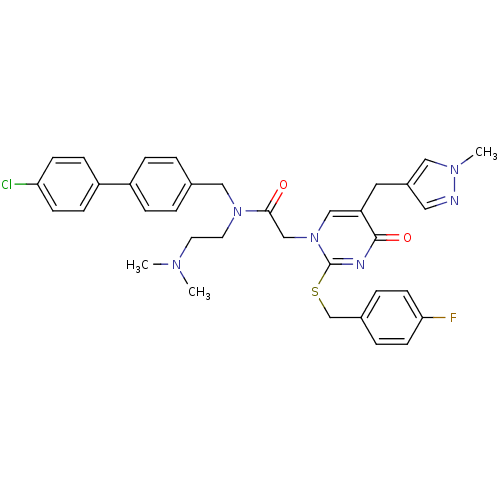
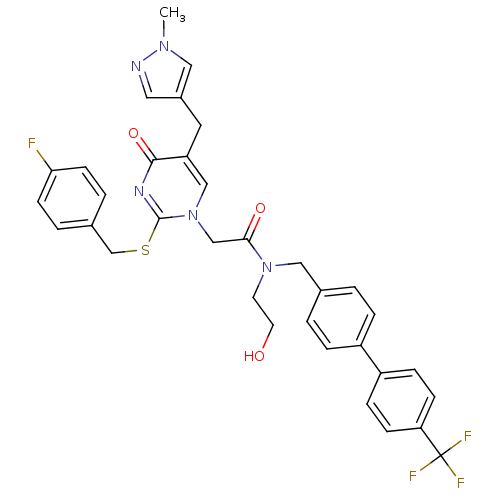
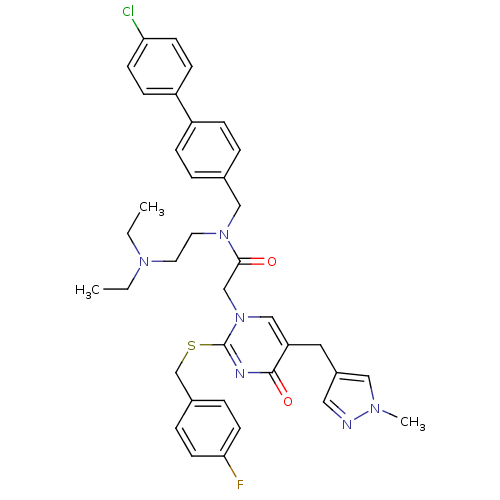
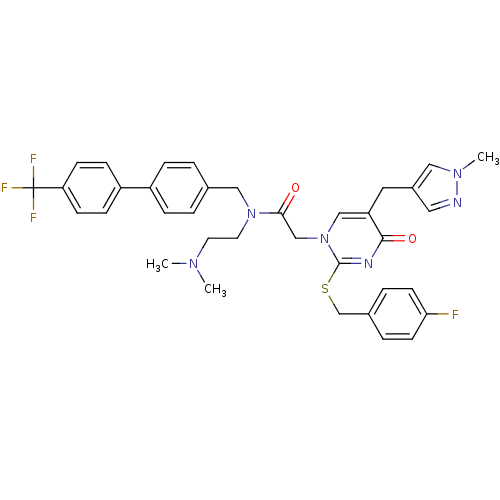
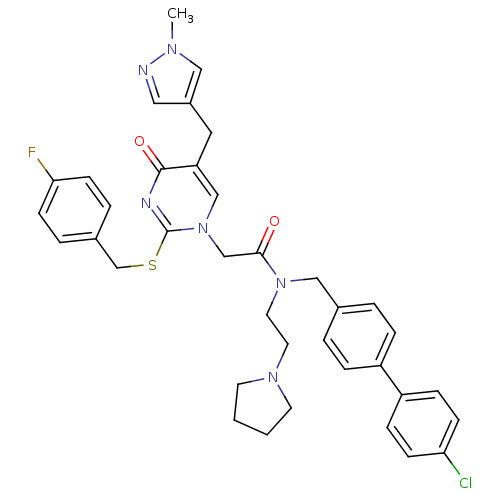
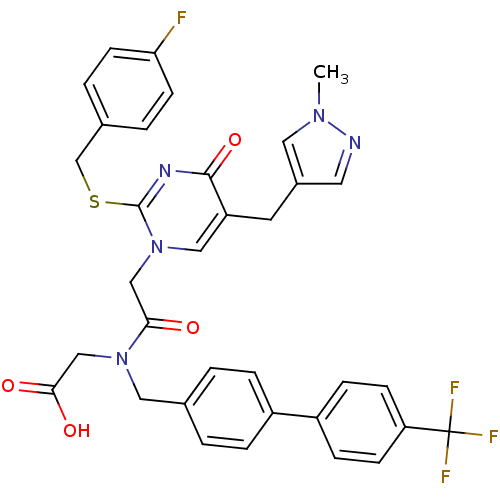
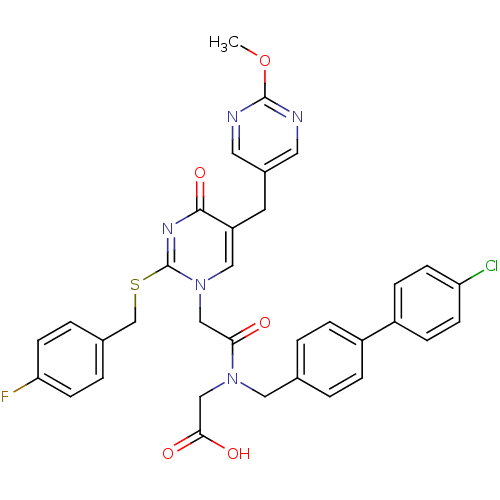
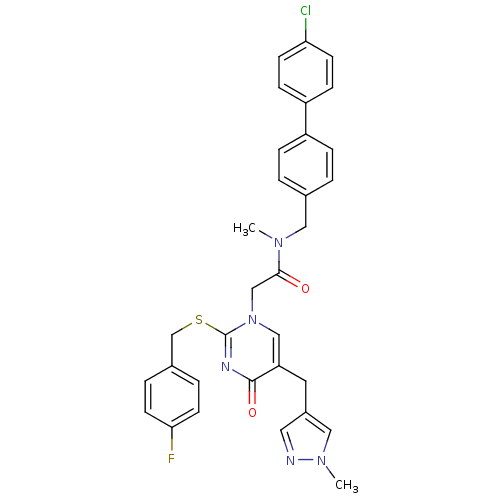
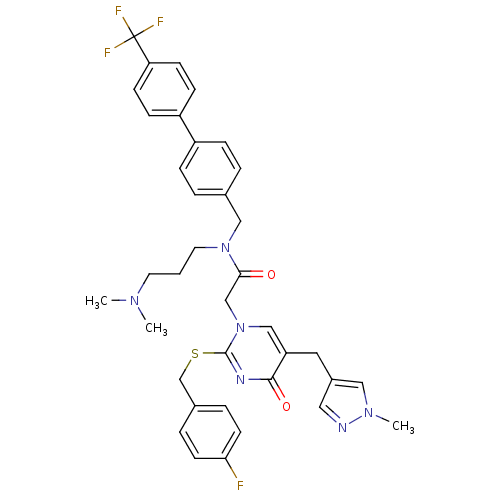
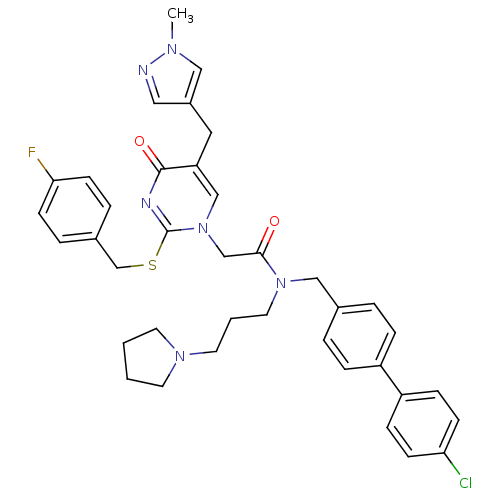
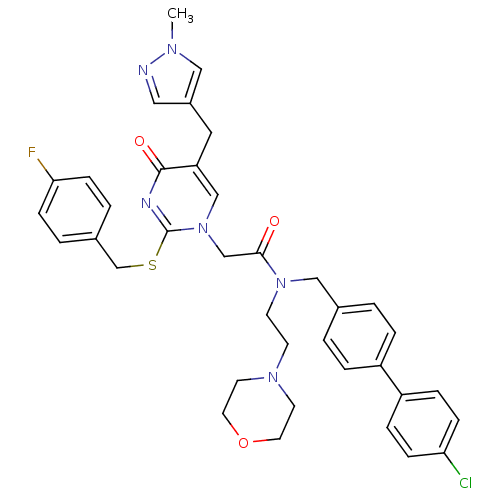
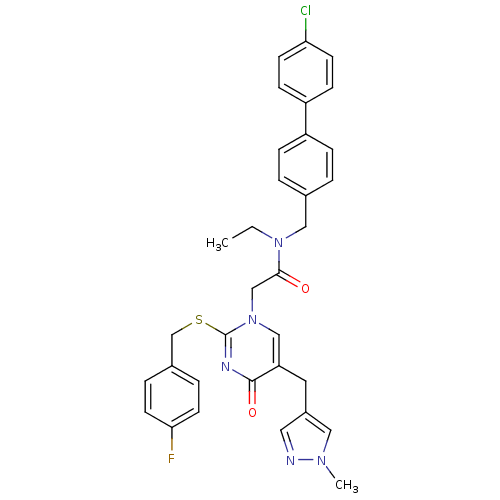
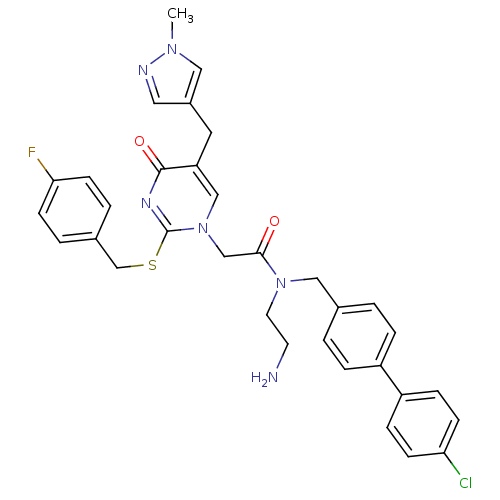
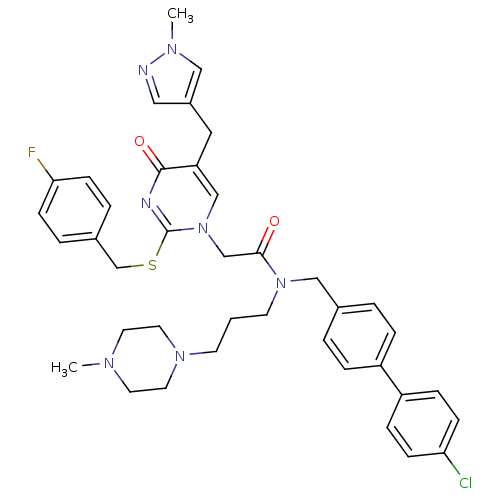
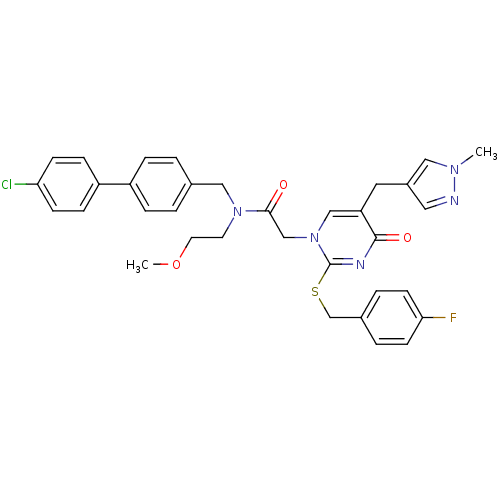
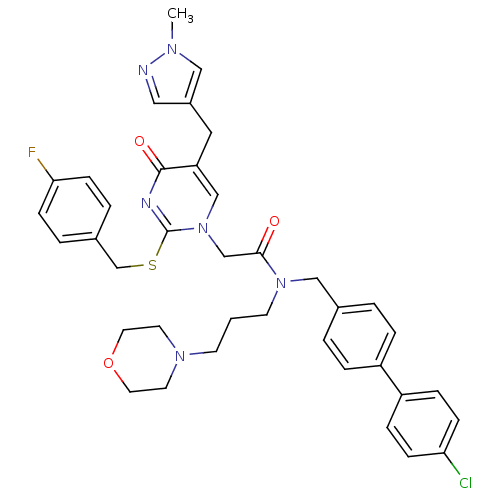
Report error Found 54 Enz. Inhib. hit(s) with all data for entry = 50012345
Affinity DataIC50: 0.00600nMAssay Description:Inhibitory concentration against Lipoprotein-Associated Phospholipase A2 (Lp-PLA2) was estimatedMore data for this Ligand-Target Pair
Affinity DataKi: 0.0300nMAssay Description:Steady state and transient kinetics to a freely reversible, non-covalently bound, human recombinant Phospholipase A2 (rhLp-PLA2) was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 0.100nMAssay Description:Steady state and transient kinetics to a freely reversible, non-covalently bound, human recombinant Phospholipase A2 (rhLp-PLA2) was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 0.120nMAssay Description:Steady state and transient kinetics to a freely reversible, non-covalently bound, human recombinant Phospholipase A2 (rhLp-PLA2) was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 0.120nMAssay Description:Steady state and transient kinetics to a freely reversible, non-covalently bound, human recombinant Phospholipase A2 (rhLp-PLA2) was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 0.170nMAssay Description:Steady state and transient kinetics to a freely reversible, non-covalently bound, human recombinant Phospholipase A2 (rhLp-PLA2) was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 0.200nMAssay Description:Steady state and transient kinetics to a freely reversible, non-covalently bound, human recombinant Phospholipase A2 (rhLp-PLA2) was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 0.200nMAssay Description:Steady state and transient kinetics to a freely reversible, non-covalently bound, human recombinant Phospholipase A2 (rhLp-PLA2) was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 0.200nMAssay Description:Steady state and transient kinetics to a freely reversible, non-covalently bound, human recombinant Phospholipase A2 (rhLp-PLA2) was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 0.200nMAssay Description:Steady state and transient kinetics to a freely reversible, non-covalently bound, human recombinant Phospholipase A2 (rhLp-PLA2) was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 0.200nMAssay Description:Steady state and transient kinetics to a freely reversible, non-covalently bound, human recombinant Phospholipase A2 (rhLp-PLA2) was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 0.200nMAssay Description:Steady state and transient kinetics to a freely reversible, non-covalently bound, human recombinant Phospholipase A2 (rhLp-PLA2) was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 0.200nMAssay Description:Steady state and transient kinetics to a freely reversible, non-covalently bound, human recombinant Phospholipase A2 (rhLp-PLA2) was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 0.300nMAssay Description:Steady state and transient kinetics to a freely reversible, non-covalently bound, human recombinant Phospholipase A2 (rhLp-PLA2) was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 0.300nMAssay Description:Steady state and transient kinetics to a freely reversible, non-covalently bound, human recombinant Phospholipase A2 (rhLp-PLA2) was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 0.350nMAssay Description:Steady state and transient kinetics to a freely reversible, non-covalently bound, human recombinant Phospholipase A2 (rhLp-PLA2) was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 0.5nMAssay Description:Steady state and transient kinetics to a freely reversible, non-covalently bound, human recombinant Phospholipase A2 (rhLp-PLA2) was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 0.5nMAssay Description:Steady state and transient kinetics to a freely reversible, non-covalently bound, human recombinant Phospholipase A2 (rhLp-PLA2) was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 0.600nMAssay Description:Steady state and transient kinetics to a freely reversible, non-covalently bound, human recombinant Phospholipase A2 (rhLp-PLA2) was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 0.600nMAssay Description:Steady state and transient kinetics to a freely reversible, non-covalently bound, human recombinant Phospholipase A2 (rhLp-PLA2) was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 0.600nMAssay Description:Steady state and transient kinetics to a freely reversible, non-covalently bound, human recombinant Phospholipase A2 (rhLp-PLA2) was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 0.700nMAssay Description:Steady state and transient kinetics to a freely reversible, non-covalently bound, human recombinant Phospholipase A2 (rhLp-PLA2) was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 0.700nMAssay Description:Steady state and transient kinetics to a freely reversible, non-covalently bound, human recombinant Phospholipase A2 (rhLp-PLA2) was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 0.800nMAssay Description:Steady state and transient kinetics to a freely reversible, non-covalently bound, human recombinant Phospholipase A2 (rhLp-PLA2) was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Steady state and transient kinetics to a freely reversible, non-covalently bound, human recombinant Phospholipase A2 (rhLp-PLA2) was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Steady state and transient kinetics to a freely reversible, non-covalently bound, human recombinant Phospholipase A2 (rhLp-PLA2) was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Steady state and transient kinetics to a freely reversible, non-covalently bound, human recombinant Phospholipase A2 (rhLp-PLA2) was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Steady state and transient kinetics to a freely reversible, non-covalently bound, human recombinant Phospholipase A2 (rhLp-PLA2) was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Steady state and transient kinetics to a freely reversible, non-covalently bound, human recombinant Phospholipase A2 (rhLp-PLA2) was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 1.40nMAssay Description:Steady state and transient kinetics to a freely reversible, non-covalently bound, human recombinant Phospholipase A2 (rhLp-PLA2) was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 1.5nMAssay Description:Steady state and transient kinetics to a freely reversible, non-covalently bound, human recombinant Phospholipase A2 (rhLp-PLA2) was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:Steady state and transient kinetics to a freely reversible, non-covalently bound, human recombinant Phospholipase A2 (rhLp-PLA2) was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:Steady state and transient kinetics to a freely reversible, non-covalently bound, human recombinant Phospholipase A2 (rhLp-PLA2) was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 3nMAssay Description:Inhibition of the Phospholipase A2 (Lp-PLA2) enzyme in whole human plasmaMore data for this Ligand-Target Pair
Affinity DataIC50: 4.5nMAssay Description:Steady state and transient kinetics to a freely reversible, non-covalently bound, human recombinant Phospholipase A2 (rhLp-PLA2) was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Steady state and transient kinetics to a freely reversible, non-covalently bound, human recombinant Phospholipase A2 (rhLp-PLA2) was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 500nMAssay Description:Inhibition of cytochrome P450 3A4More data for this Ligand-Target Pair
Affinity DataIC50: 500nMAssay Description:Inhibition of cytochrome P450 3A4More data for this Ligand-Target Pair
Affinity DataIC50: 2.00E+3nMAssay Description:Inhibition of cytochrome P450 3A4More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+3nMAssay Description:Inhibition of cytochrome P450 3A4More data for this Ligand-Target Pair
Affinity DataIC50: 6.00E+3nMAssay Description:Inhibition of cytochrome P450 3A4More data for this Ligand-Target Pair
Affinity DataIC50: 8.00E+3nMAssay Description:Inhibition of cytochrome P450 3A4More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibition of cytochrome P450 3A4More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibition of cytochrome P450 3A4More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibition of cytochrome P450 3A4More data for this Ligand-Target Pair
Affinity DataIC50: 1.50E+4nMAssay Description:Inhibition of cytochrome P450 3A4More data for this Ligand-Target Pair
Affinity DataIC50: 1.70E+4nMAssay Description:Inhibition of cytochrome P450 3A4More data for this Ligand-Target Pair
Affinity DataIC50: 1.90E+4nMAssay Description:Inhibition of cytochrome P450 3A4More data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+4nMAssay Description:Inhibition of cytochrome P450 3A4More data for this Ligand-Target Pair
Affinity DataIC50: 3.10E+4nMAssay Description:Inhibition of cytochrome P450 3A4More data for this Ligand-Target Pair