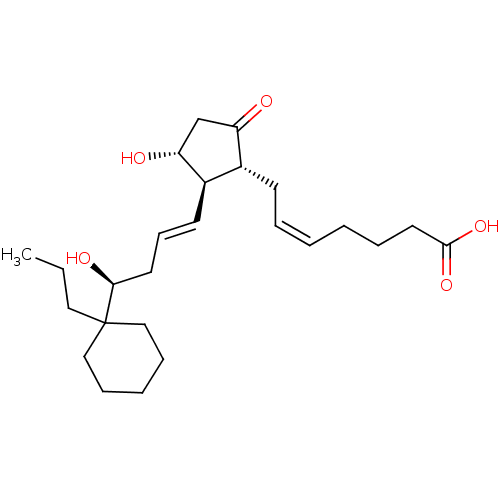
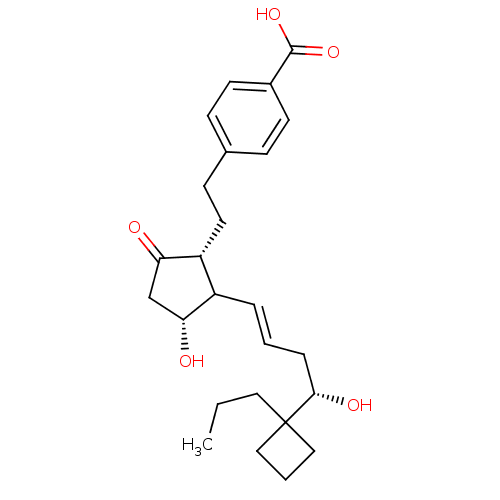
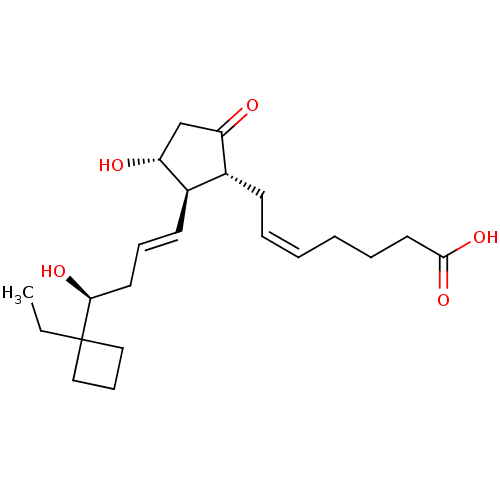
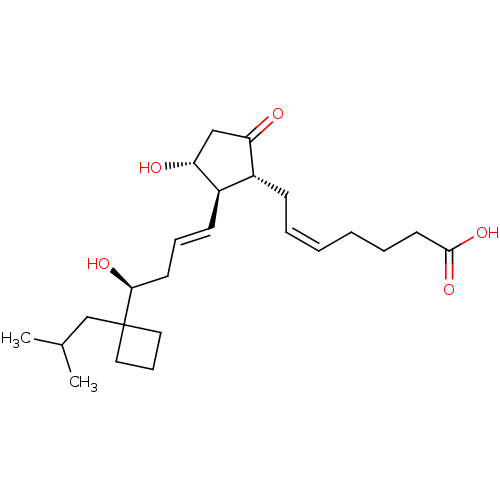
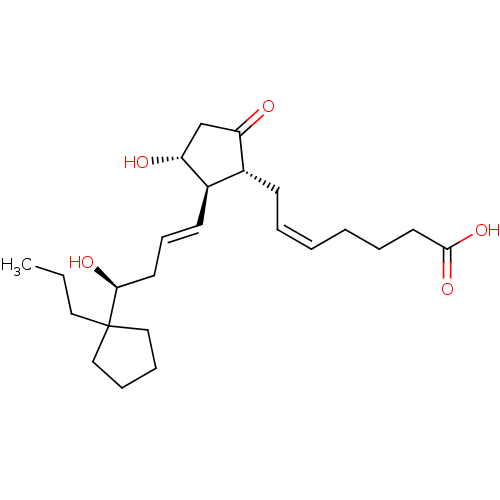
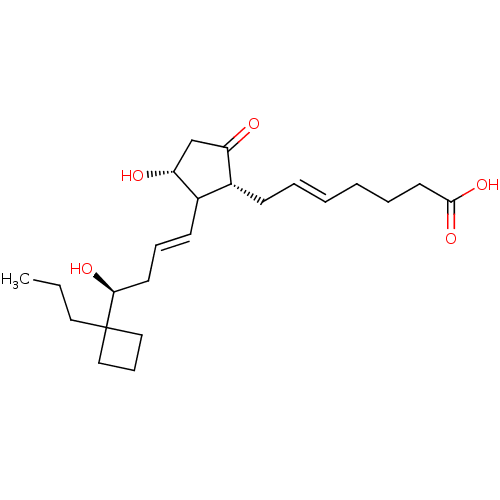
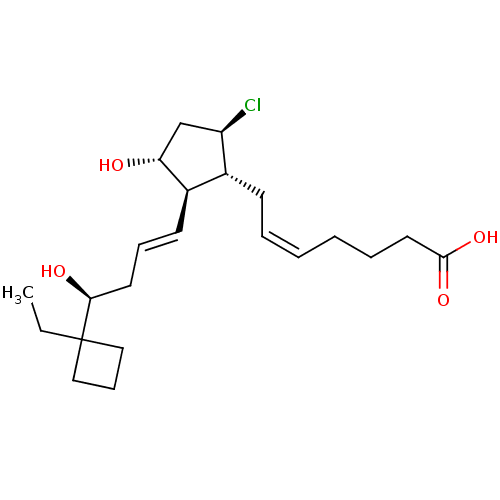
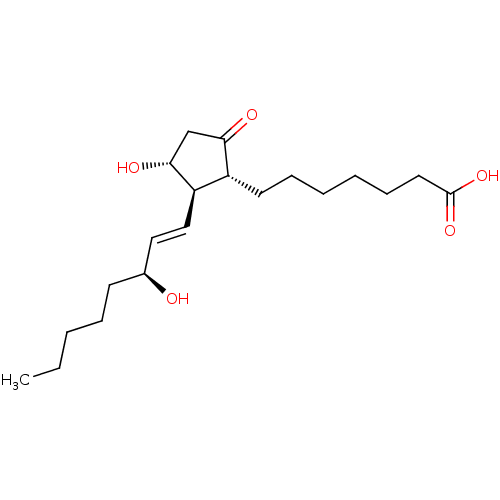
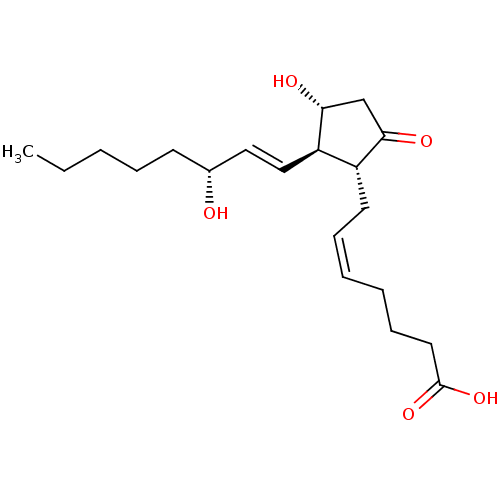
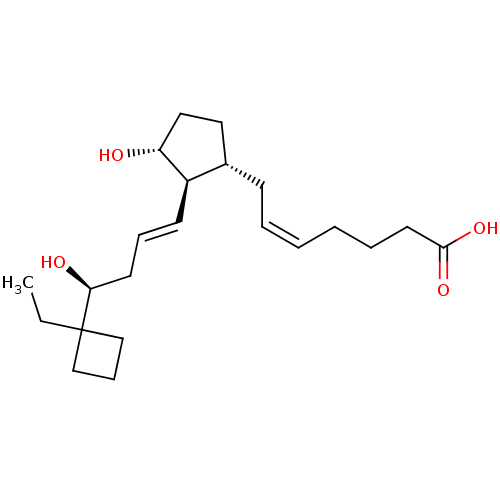
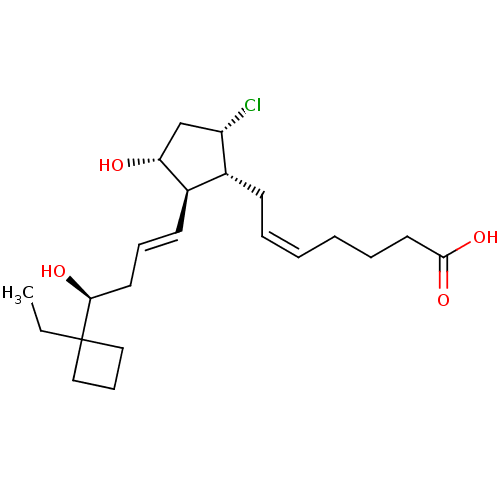
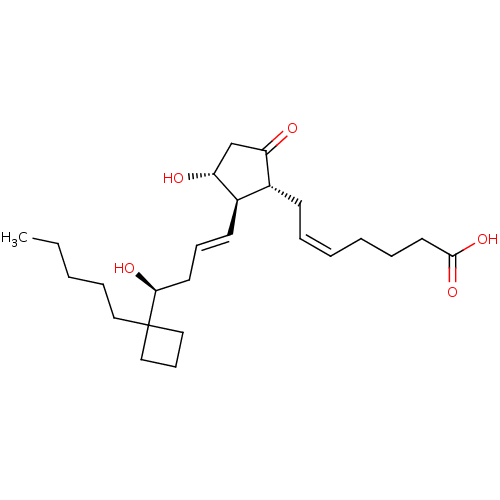
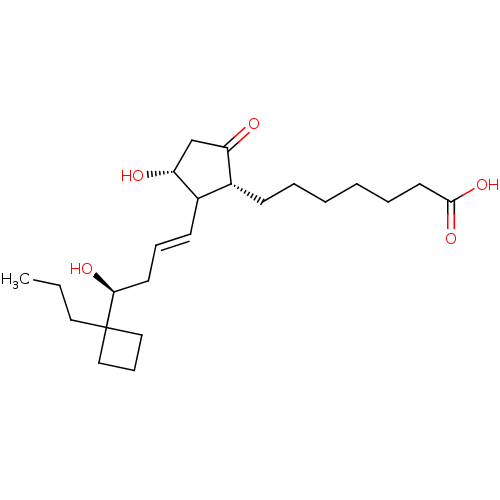
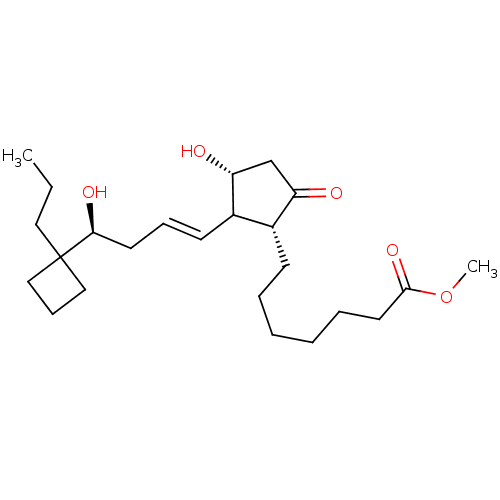
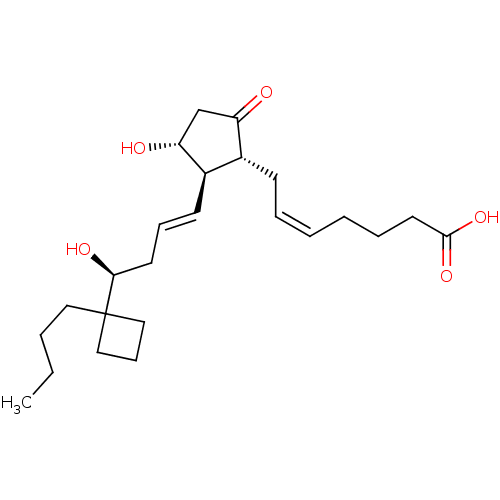
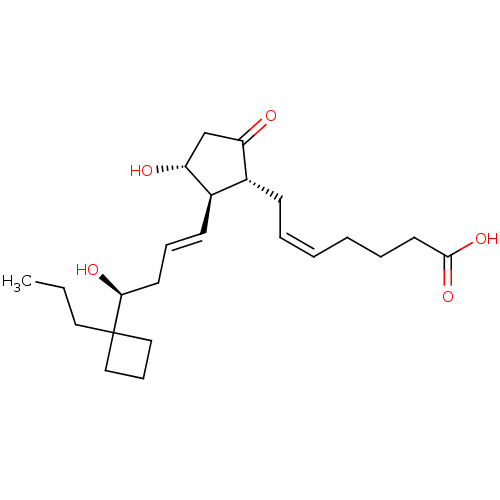
Report error Found 107 Enz. Inhib. hit(s) with all data for entry = 50011059
Affinity DataKi: >0.000100nMAssay Description:Binding affinity towards mouse Prostanoid EP3 receptor in CHO cells.More data for this Ligand-Target Pair
Affinity DataKi: >0.000100nMAssay Description:Effective concentration which increases intracellular c-AMP production in human Prostanoid IP receptorMore data for this Ligand-Target Pair
Affinity DataKi: >0.000100nMAssay Description:Binding affinity towards mouse Prostanoid EP3 receptor in CHO cells.More data for this Ligand-Target Pair
Affinity DataKi: >0.000100nMAssay Description:Binding affinity towards mouse Prostanoid EP3 receptor in CHO cells.More data for this Ligand-Target Pair
Affinity DataKi: >0.000100nMAssay Description:Binding affinity towards mouse Prostanoid EP3 receptor in CHO cells.More data for this Ligand-Target Pair
Affinity DataKi: >0.000100nMAssay Description:Binding affinity towards mouse Prostanoid EP3 receptor in CHO cells.More data for this Ligand-Target Pair
Affinity DataKi: >0.000100nMAssay Description:Binding affinity towards mouse Prostanoid EP3 receptor in CHO cells.More data for this Ligand-Target Pair
Affinity DataKi: >0.000100nMAssay Description:Effective concentration which increases intracellular c-AMP production in human Prostanoid IP receptorMore data for this Ligand-Target Pair
Affinity DataKi: >0.000100nMAssay Description:Effective concentration which increases intracellular c-AMP production in human Prostanoid IP receptorMore data for this Ligand-Target Pair
Affinity DataKi: 0.740nMAssay Description:Compound was evaluated for its competitive binding affinity towards human Prostanoid EP2 receptor in CHO cells expressing prostanoid receptorMore data for this Ligand-Target Pair
Affinity DataEC50: 1.80nMAssay Description:Effective concentration which increases intracellular c-AMP production in human Prostanoid IP receptorMore data for this Ligand-Target Pair
Affinity DataEC50: 2.10nMAssay Description:Effective concentration which increases intracellular c-AMP production in mouse Prostanoid EP2 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 2.20nMAssay Description:Binding affinity towards mouse Prostanoid EP2 receptor expressed in CHO cells.More data for this Ligand-Target Pair
Affinity DataEC50: 2.60nMAssay Description:Effective concentration which increases intracellular c-AMP production in mouse Prostanoid EP2 receptorMore data for this Ligand-Target Pair
Affinity DataEC50: 2.80nMAssay Description:Effective concentration which increases intracellular c-AMP production in mouse Prostanoid EP2 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 3.10nMAssay Description:Binding affinity towards mouse Prostanoid EP4 receptor in CHO cells.More data for this Ligand-Target Pair
Affinity DataKi: 3.30nMAssay Description:Binding affinity towards mouse Prostanoid EP4 receptor in CHO cells.More data for this Ligand-Target Pair
Affinity DataKi: 5nMAssay Description:Binding affinity towards mouse Prostanoid EP3 receptor in CHO cells.More data for this Ligand-Target Pair
Affinity DataKi: 5nMAssay Description:Binding affinity towards mouse Prostanoid EP3 receptor in CHO cells.More data for this Ligand-Target Pair
Affinity DataKi: 11nMAssay Description:Binding affinity towards mouse Prostanoid EP2 receptor expressed in CHO cells.More data for this Ligand-Target Pair
Affinity DataEC50: 11nMAssay Description:Effective concentration which increases intracellular c-AMP production in mouse Prostanoid EP2 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:Binding affinity towards mouse Prostanoid EP2 receptor expressed in CHO cells.More data for this Ligand-Target Pair
Affinity DataKi: 18nMAssay Description:Binding affinity towards mouse Prostanoid EP1 receptor in CHO cells.More data for this Ligand-Target Pair
Affinity DataKi: 20nMAssay Description:Binding affinity towards mouse Prostanoid EP2 receptor expressed in CHO cells.More data for this Ligand-Target Pair
Affinity DataEC50: 25nMAssay Description:Effective concentration which increases intracellular c-AMP production in human Prostanoid IP receptorMore data for this Ligand-Target Pair
Affinity DataKi: 25nMAssay Description:Binding affinity towards mouse Prostanoid EP2 receptor expressed in CHO cells.More data for this Ligand-Target Pair
Affinity DataKi: 30nMAssay Description:Binding affinity towards mouse Prostanoid EP2 receptor expressed in CHO cells.More data for this Ligand-Target Pair
Affinity DataEC50: 32nMAssay Description:Effective concentration which increases intracellular c-AMP production in mouse Prostanoid EP2 receptorMore data for this Ligand-Target Pair
Affinity DataEC50: 33nMAssay Description:Effective concentration which increases intracellular c-AMP production in mouse EP2- receptorMore data for this Ligand-Target Pair
Affinity DataEC50: 37nMAssay Description:Effective concentration which increases intracellular c-AMP production in human Prostanoid IP receptorMore data for this Ligand-Target Pair
Affinity DataKi: 38nMAssay Description:Binding affinity towards mouse Prostanoid EP2 receptor expressed in CHO cells.More data for this Ligand-Target Pair
Affinity DataEC50: 39nMAssay Description:Effective concentration which increases intracellular c-AMP production in mouse Prostanoid EP2 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 40nMAssay Description:Binding affinity towards mouse Prostanoid EP2 receptor expressed in CHO cells.More data for this Ligand-Target Pair
Affinity DataKi: 43nMAssay Description:Binding affinity towards mouse Prostanoid EP2 receptor expressed in CHO cells.More data for this Ligand-Target Pair
Affinity DataEC50: 43nMAssay Description:Effective concentration which increases intracellular c-AMP production in mouse Prostanoid EP2 receptorMore data for this Ligand-Target Pair
Affinity DataEC50: 45nMAssay Description:Effective concentration which increases intracellular c-AMP production in mouse Prostanoid EP2 receptorMore data for this Ligand-Target Pair
Affinity DataEC50: 54nMAssay Description:Effective concentration which increases intracellular c-AMP production in mouse Prostanoid EP2 receptorMore data for this Ligand-Target Pair
Affinity DataEC50: 71nMAssay Description:Effective concentration which increases intracellular c-AMP production in mouse Prostanoid EP2 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 73nMAssay Description:Binding affinity towards mouse Prostanoid EP2 receptor expressed in CHO cells.More data for this Ligand-Target Pair
Affinity DataKi: 87nMAssay Description:Binding affinity towards mouse Prostanoid EP2 receptor expressed in CHO cells.More data for this Ligand-Target Pair
Affinity DataKi: 92nMAssay Description:Binding affinity towards mouse Prostanoid EP2 receptor expressed in CHO cells.More data for this Ligand-Target Pair
Affinity DataKi: 100nMAssay Description:Binding affinity towards mouse Prostanoid EP1 receptor in CHO cells.More data for this Ligand-Target Pair
Affinity DataEC50: 130nMAssay Description:Effective concentration which increases intracellular c-AMP production in mouse Prostanoid EP2 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 150nMAssay Description:Binding affinity towards human Prostanoid IP receptor in CHO cells.More data for this Ligand-Target Pair
Affinity DataEC50: 220nMAssay Description:Effective concentration which increases intracellular c-AMP production in mouse Prostanoid EP2 receptorMore data for this Ligand-Target Pair
Affinity DataEC50: 260nMAssay Description:Effective concentration which increases intracellular c-AMP production in human Prostanoid IP receptorMore data for this Ligand-Target Pair
Affinity DataKi: 330nMAssay Description:Binding affinity towards mouse Prostanoid EP2 receptor expressed in CHO cells.More data for this Ligand-Target Pair
Affinity DataKi: 370nMAssay Description:Binding affinity towards mouse Prostanoid EP2 receptor expressed in CHO cells.More data for this Ligand-Target Pair
Affinity DataEC50: 580nMAssay Description:Effective concentration which increases intracellular c-AMP production in mouse Prostanoid EP2 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 780nMAssay Description:Binding affinity towards mouse Prostanoid EP1 receptor in CHO cells.More data for this Ligand-Target Pair