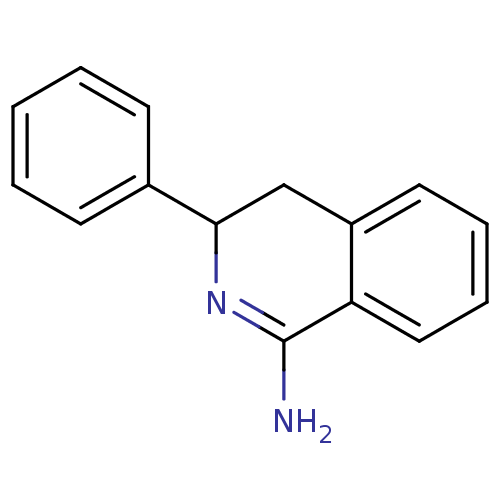
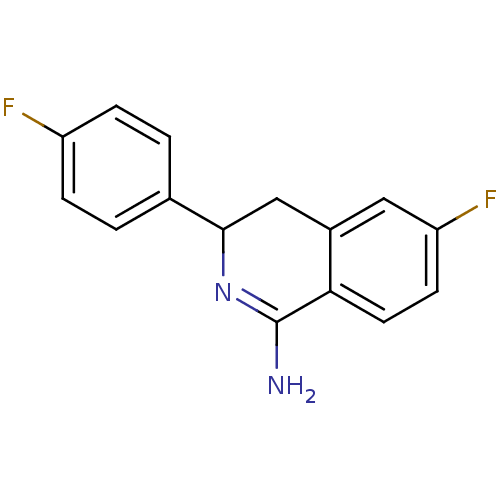
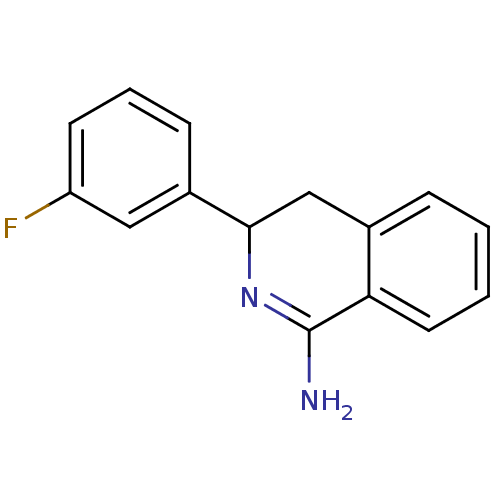
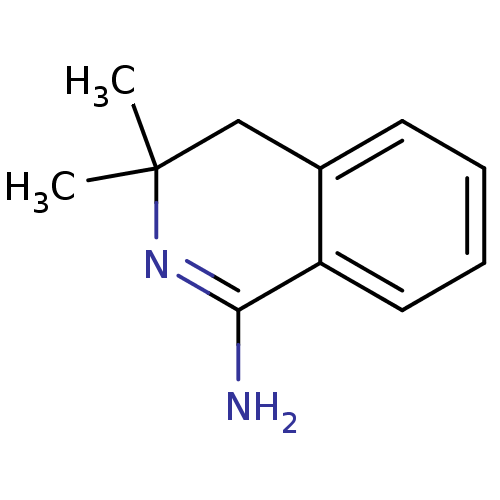
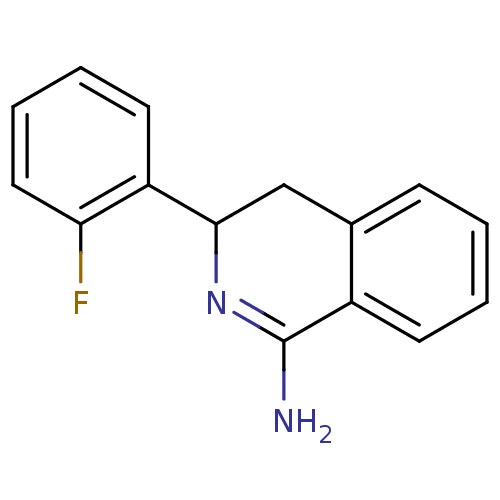
Report error Found 73 Enz. Inhib. hit(s) with all data for entry = 50010803
Affinity DataIC50: 100nMAssay Description:Inhibition of conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by neuronal NOS (n NOS) from rat cerebellumMore data for this Ligand-Target Pair
Affinity DataIC50: 130nMAssay Description:Inhibitory concentration against nitric oxide synthesis in intact DLD-1 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Inhibition of conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by neuronal NOS (n NOS) from rat cerebellumMore data for this Ligand-Target Pair
Affinity DataIC50: 160nMAssay Description:Ability to inhibit conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by endothelial NOS (e NOS) from HUVEC cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:Ability to inhibit conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by endothelial NOS (e NOS) from HUVEC cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:Ability to inhibit conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by inducible NOS (i NOS) from human DLD-1 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 400nMAssay Description:Inhibition of conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by neuronal NOS (n NOS) from rat cerebellumMore data for this Ligand-Target Pair
Affinity DataIC50: 400nMAssay Description:Inhibition of conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by neuronal NOS (n NOS) from rat cerebellumMore data for this Ligand-Target Pair
Affinity DataIC50: 550nMAssay Description:Inhibitory concentration against nitric oxide synthesis in intact DLD-1 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 900nMAssay Description:Inhibition of conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by neuronal NOS (n NOS) from rat cerebellumMore data for this Ligand-Target Pair
Affinity DataIC50: 900nMAssay Description:Ability to inhibit conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by endothelial NOS (e NOS) from HUVEC cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 1.20E+3nMAssay Description:Inhibitory concentration against nitric oxide synthesis in intact DLD-1 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 1.30E+3nMAssay Description:Inhibitory concentration against nitric oxide synthesis in intact DLD-1 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 1.50E+3nMAssay Description:Inhibition of conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by neuronal NOS (n NOS) from rat cerebellumMore data for this Ligand-Target Pair
Affinity DataIC50: 1.60E+3nMAssay Description:Inhibition of conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by neuronal NOS (n NOS) from rat cerebellumMore data for this Ligand-Target Pair
Affinity DataIC50: 2.00E+3nMAssay Description:Inhibition of conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by neuronal NOS (n NOS) from rat cerebellumMore data for this Ligand-Target Pair
Affinity DataIC50: 2.00E+3nMAssay Description:Inhibition of conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by neuronal NOS (n NOS) from rat cerebellumMore data for this Ligand-Target Pair
Affinity DataIC50: 2.00E+3nMAssay Description:Ability to inhibit conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by endothelial NOS (e NOS) from HUVEC cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 2.00E+3nMAssay Description:Inhibition of conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by neuronal NOS (n NOS) from rat cerebellumMore data for this Ligand-Target Pair
Affinity DataIC50: 2.40E+3nMAssay Description:Ability to inhibit conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by endothelial NOS (e NOS) from HUVEC cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 2.70E+3nMAssay Description:Inhibitory concentration against nitric oxide synthesis in intact DLD-1 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 2.70E+3nMAssay Description:Ability to inhibit conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by endothelial NOS (e NOS) from HUVEC cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 2.70E+3nMAssay Description:Inhibitory concentration against nitric oxide synthesis in intact DLD-1 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+3nMAssay Description:Inhibition of conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by neuronal NOS (n NOS) from rat cerebellumMore data for this Ligand-Target Pair
Affinity DataIC50: 3.60E+3nMAssay Description:Ability to inhibit conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by endothelial NOS (e NOS) from HUVEC cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 4.00E+3nMAssay Description:Ability to inhibit conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by endothelial NOS (e NOS) from HUVEC cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 4.20E+3nMAssay Description:Ability to inhibit conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by inducible NOS (i NOS) from human DLD-1 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 4.50E+3nMAssay Description:Ability to inhibit conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by inducible NOS (i NOS) from human DLD-1 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 4.70E+3nMAssay Description:Inhibitory concentration against nitric oxide synthesis in intact DLD-1 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+3nMAssay Description:Ability to inhibit conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by endothelial NOS (e NOS) from HUVEC cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+3nMAssay Description:Inhibition of conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by neuronal NOS (n NOS) from rat cerebellumMore data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+3nMAssay Description:Ability to inhibit conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by endothelial NOS (e NOS) from HUVEC cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 5.50E+3nMAssay Description:Inhibitory concentration against nitric oxide synthesis in intact DLD-1 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 6.00E+3nMAssay Description:Inhibition of conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by neuronal NOS (n NOS) from rat cerebellumMore data for this Ligand-Target Pair
Affinity DataIC50: 6.00E+3nMAssay Description:Ability to inhibit conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by endothelial NOS (e NOS) from HUVEC cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 8.00E+3nMAssay Description:Inhibition of conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by neuronal NOS (n NOS) from rat cerebellumMore data for this Ligand-Target Pair
Affinity DataIC50: 8.00E+3nMAssay Description:Ability to inhibit conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by endothelial NOS (e NOS) from HUVEC cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 9.00E+3nMAssay Description:Ability to inhibit conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by endothelial NOS (e NOS) from HUVEC cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 9.00E+3nMAssay Description:Inhibitory concentration against nitric oxide synthesis in intact DLD-1 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 9.00E+3nMAssay Description:Inhibition of conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by neuronal NOS (n NOS) from rat cerebellumMore data for this Ligand-Target Pair
Affinity DataIC50: 9.00E+3nMAssay Description:Ability to inhibit conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by endothelial NOS (e NOS) from HUVEC cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibition of conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by neuronal NOS (n NOS) from rat cerebellumMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibition of conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by neuronal NOS (n NOS) from rat cerebellumMore data for this Ligand-Target Pair
Affinity DataIC50: 1.20E+4nMAssay Description:Ability to inhibit conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by endothelial NOS (e NOS) from HUVEC cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 1.20E+4nMAssay Description:Inhibition of conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by neuronal NOS (n NOS) from rat cerebellumMore data for this Ligand-Target Pair
Affinity DataIC50: 1.30E+4nMAssay Description:Inhibition of conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by neuronal NOS (n NOS) from rat cerebellumMore data for this Ligand-Target Pair
Affinity DataIC50: 1.40E+4nMAssay Description:Inhibitory concentration against nitric oxide synthesis in intact DLD-1 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 1.60E+4nMAssay Description:Inhibition of conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by neuronal NOS (n NOS) from rat cerebellumMore data for this Ligand-Target Pair
Affinity DataIC50: 1.80E+4nMAssay Description:Inhibitory concentration against nitric oxide synthesis in intact DLD-1 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 1.90E+4nMAssay Description:Ability to inhibit conversion of [3H]L-Arg to [3H]L-citrulline catalyzed by endothelial NOS (e NOS) from HUVEC cellsMore data for this Ligand-Target Pair
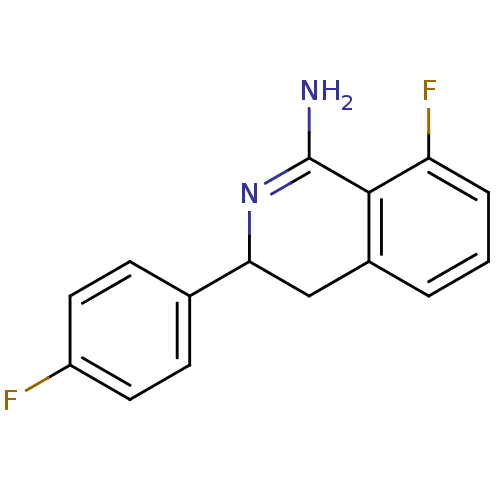
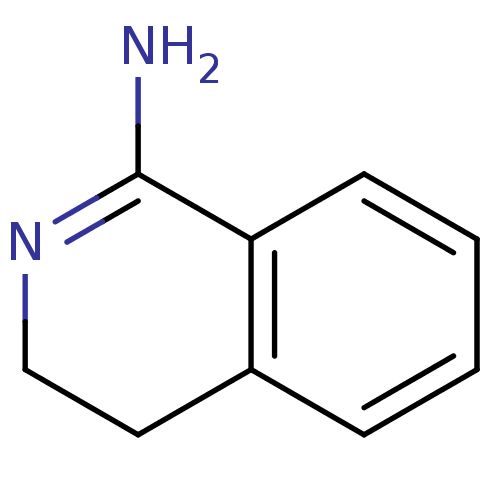
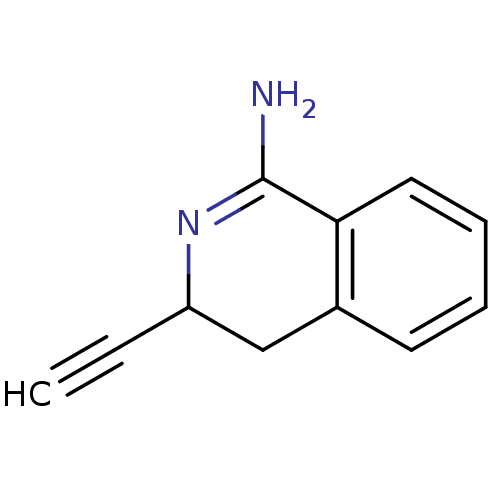
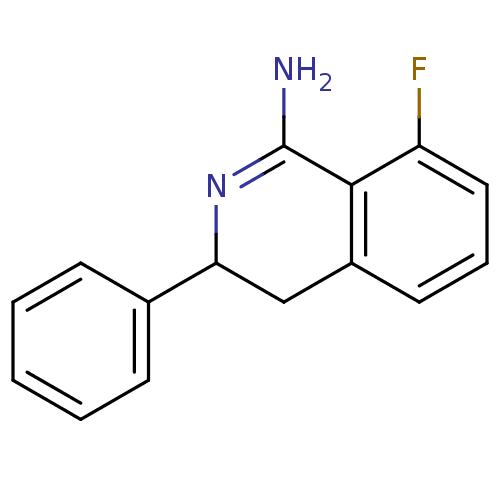
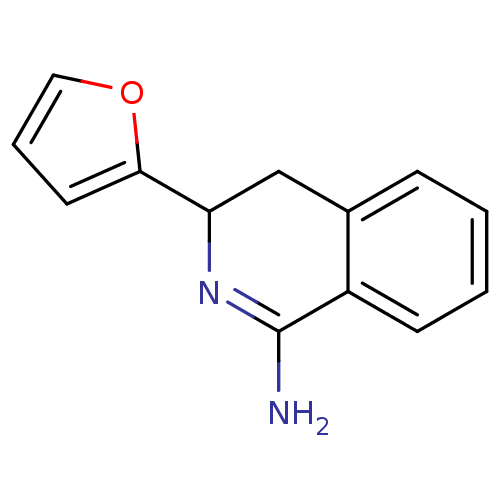
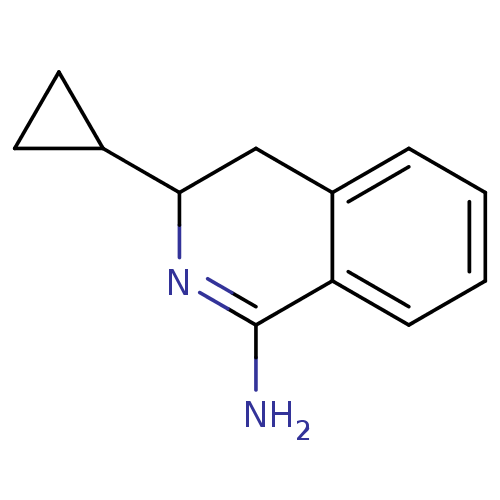
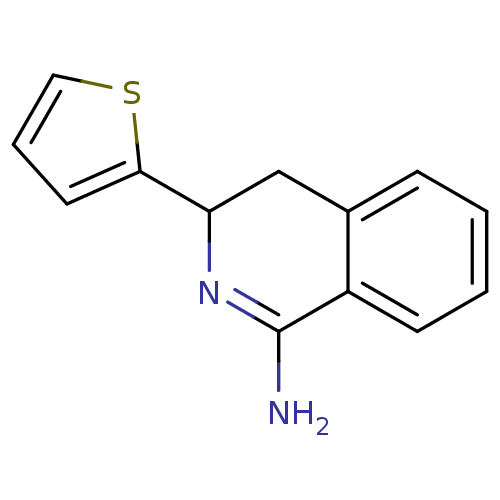
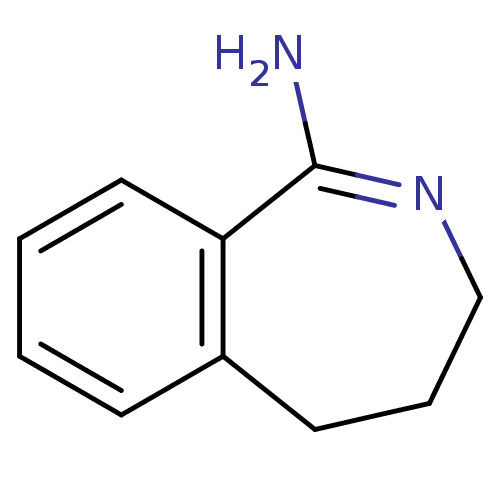
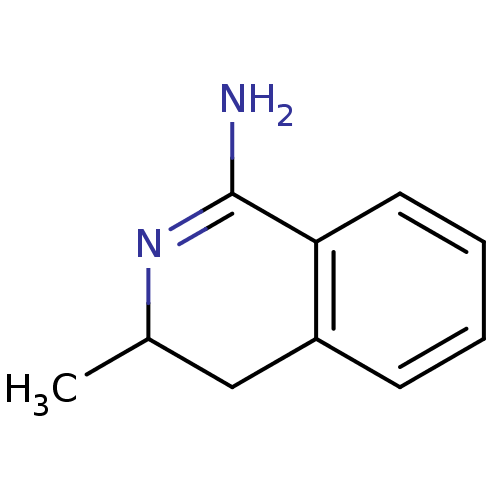
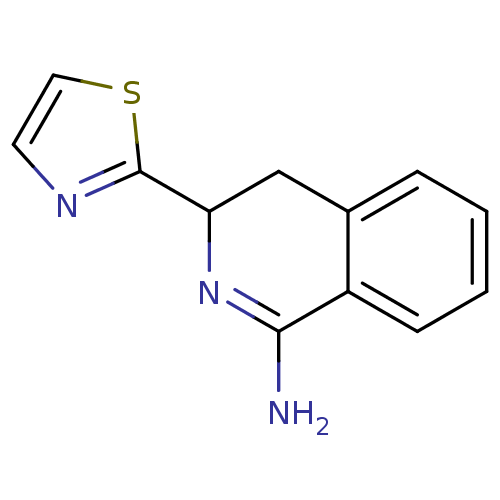
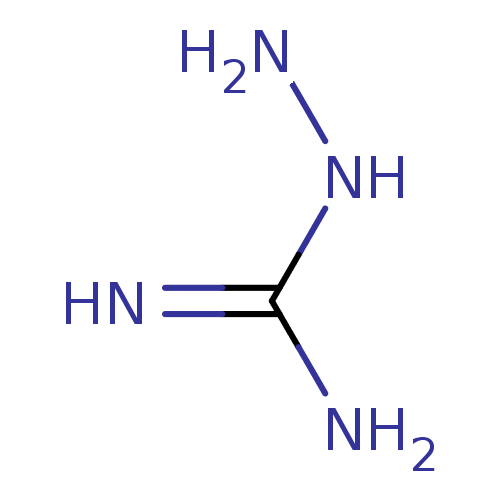
 3D Structure (crystal)
3D Structure (crystal)