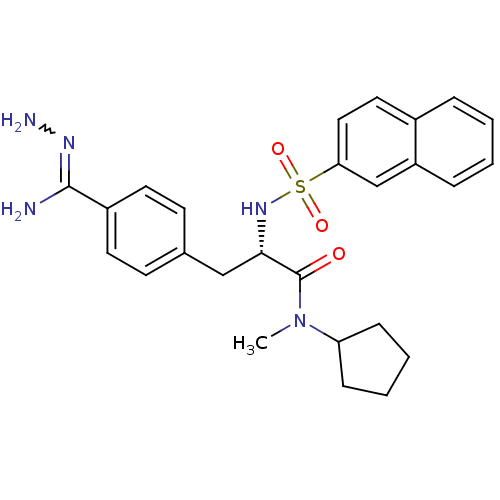
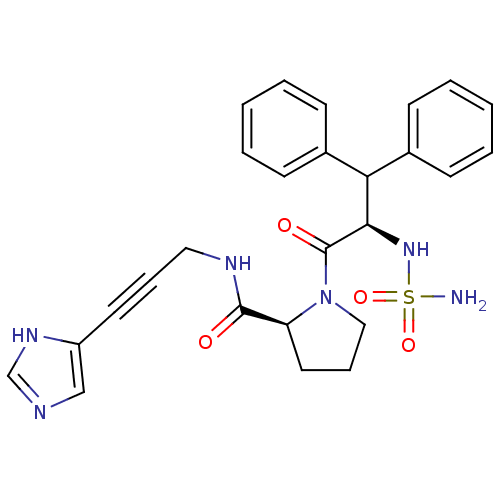
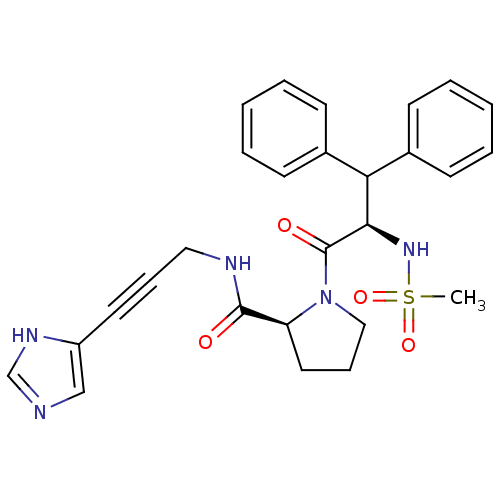
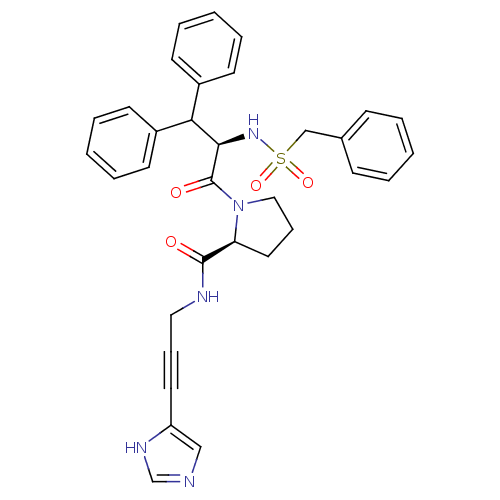
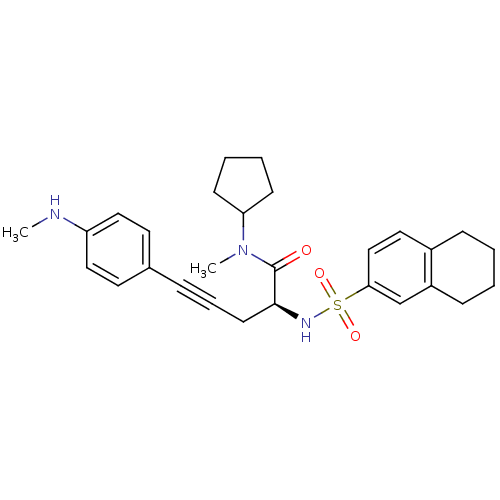
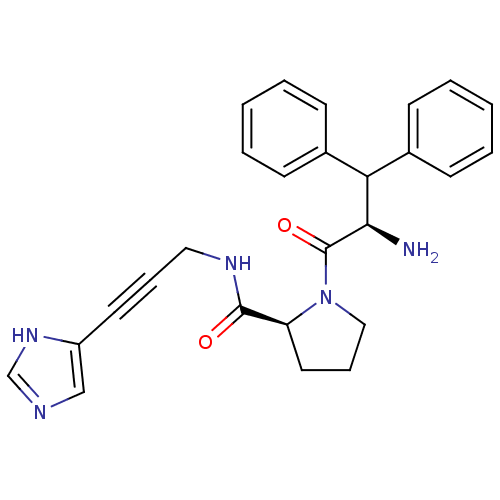
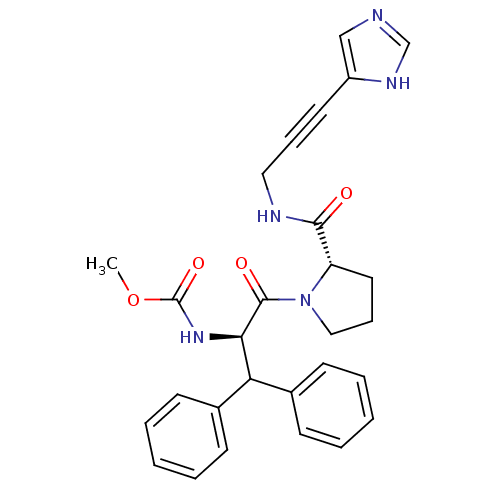
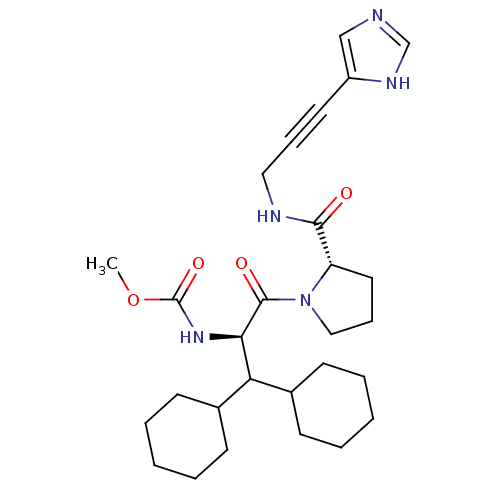
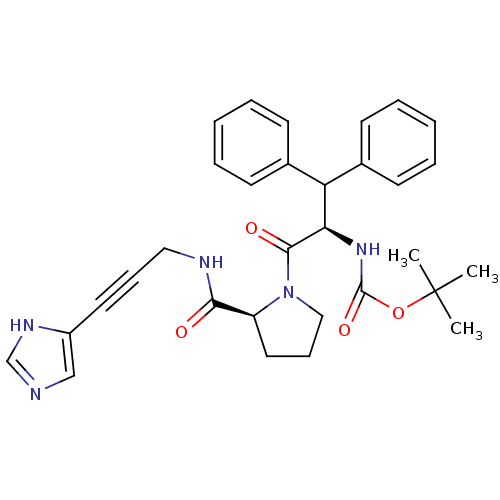
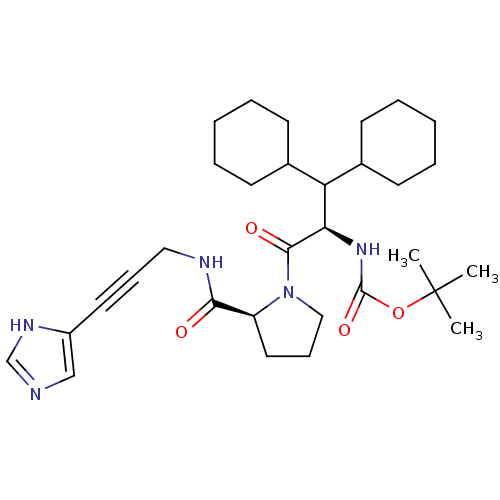
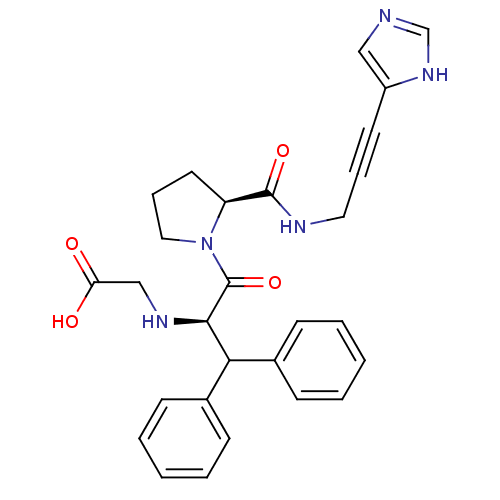
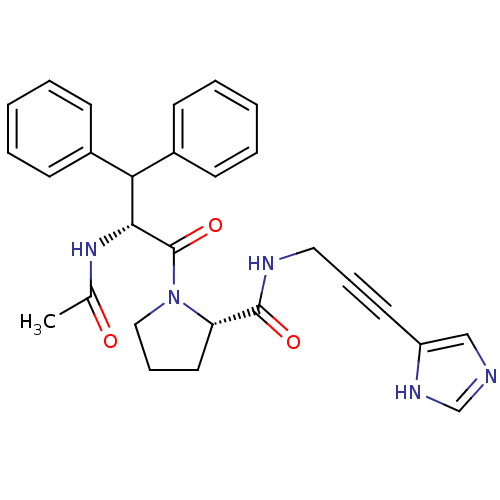
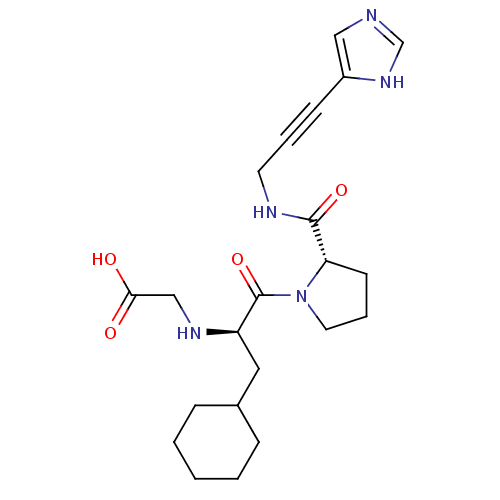
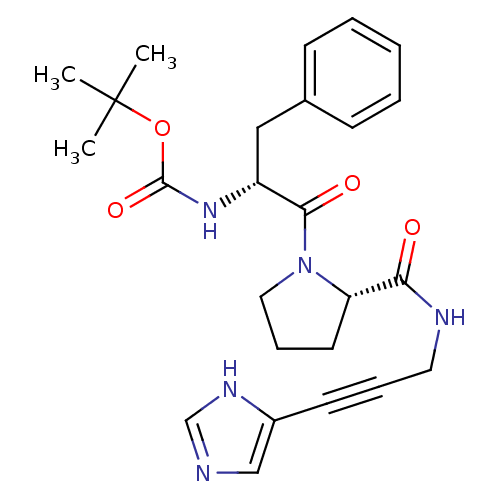
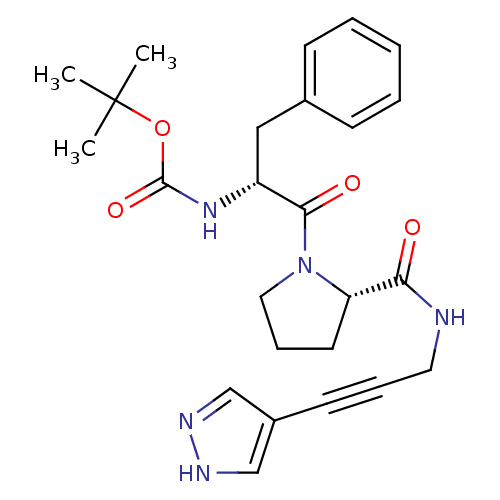
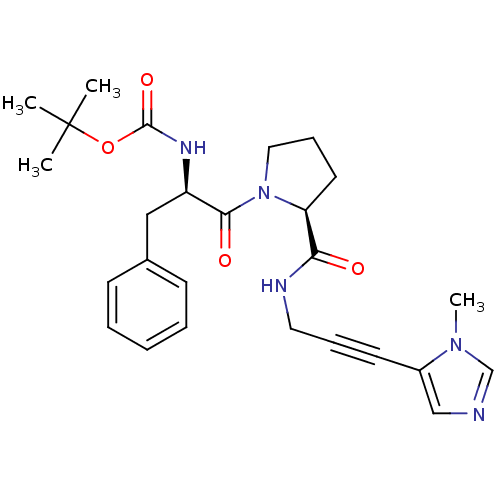
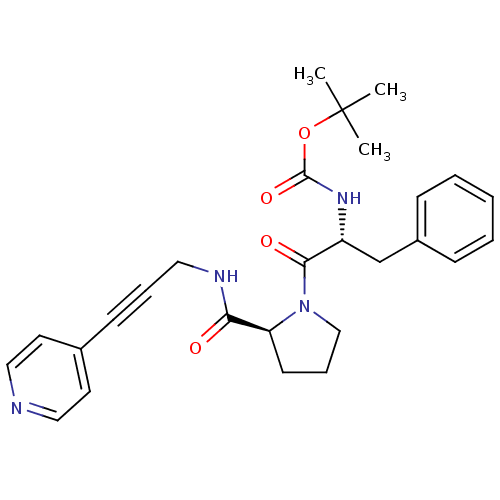
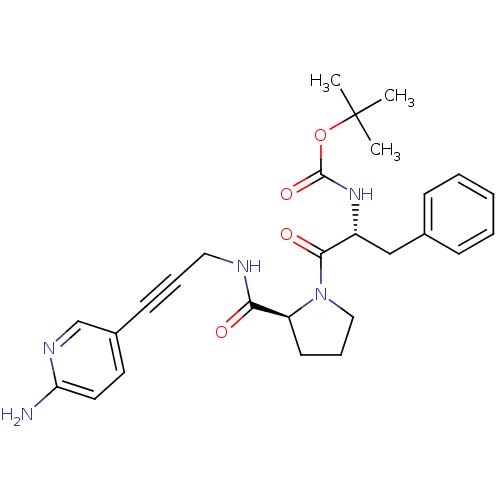
Report error Found 28 Enz. Inhib. hit(s) with all data for entry = 50010470
Affinity DataKi: 0.400nMAssay Description:In vitro inhibition of human thrombin.More data for this Ligand-Target Pair
Affinity DataKi: 0.800nMAssay Description:In vitro inhibition of human thrombin.More data for this Ligand-Target Pair
Affinity DataKi: 2.60nMAssay Description:In vitro inhibition of human thrombin.More data for this Ligand-Target Pair
Affinity DataKi: 3nMAssay Description:In vitro inhibition of human thrombin.More data for this Ligand-Target Pair
Affinity DataKi: 5nMAssay Description:In vitro inhibition of human thrombin.More data for this Ligand-Target Pair
Affinity DataKi: 12nMAssay Description:In vitro inhibition of human thrombin.More data for this Ligand-Target Pair
Affinity DataKi: 14nMAssay Description:Inhibitory activity against Bovine TrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 14nMAssay Description:In vitro inhibition of human thrombin.More data for this Ligand-Target Pair
Affinity DataKi: 15nMAssay Description:In vitro inhibition of human thrombin.More data for this Ligand-Target Pair
Affinity DataKi: 18nMAssay Description:Inhibitory activity against Bovine TrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 24nMAssay Description:Inhibitory activity against Bovine TrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 24nMAssay Description:In vitro inhibition of human thrombin.More data for this Ligand-Target Pair
Affinity DataKi: 24nMAssay Description:Inhibitory activity against Bovine TrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 25nMAssay Description:Inhibitory activity against Bovine TrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 29nMAssay Description:In vitro inhibition of human thrombin.More data for this Ligand-Target Pair
Affinity DataKi: 30nMAssay Description:Inhibitory activity against Bovine TrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 30nMAssay Description:Inhibitory activity against Bovine TrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 33nMAssay Description:Inhibitory activity against Bovine TrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 44nMAssay Description:In vitro inhibition of human thrombin.More data for this Ligand-Target Pair
Affinity DataKi: 47nMAssay Description:Inhibitory activity against Bovine TrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 50nMAssay Description:Inhibitory activity against Bovine TrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 208nMAssay Description:In vitro inhibition of human thrombin.More data for this Ligand-Target Pair
Affinity DataKi: 480nMAssay Description:In vitro inhibition of human thrombin.More data for this Ligand-Target Pair
Affinity DataKi: 1.20E+3nMAssay Description:In vitro inhibition of human thrombin.More data for this Ligand-Target Pair
Affinity DataKi: 5.40E+3nMAssay Description:In vitro inhibition of human thrombin.More data for this Ligand-Target Pair
Affinity DataKi: 5.50E+3nMAssay Description:In vitro inhibition of human thrombin.More data for this Ligand-Target Pair
Affinity DataKi: >2.00E+4nMAssay Description:In vitro inhibition of human thrombin.More data for this Ligand-Target Pair
Affinity DataKi: >2.00E+4nMAssay Description:In vitro inhibition of human thrombin.More data for this Ligand-Target Pair