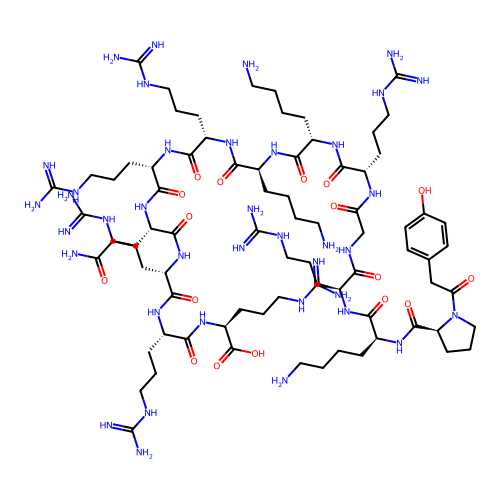
BDBM66879 (2-(4-hydroxyphenyl)acetyl)-L-prolyl-L-lysyl-L-arginylglycyl-L-arginyl-L-lysyl-L-lysyl-L-arginyl-L-arginyl-L-glutaminyl-L-arginyl-L-arginyl-L-arginine::US12479887, Compound 32
SMILES N=C(N)NCCC[C@H](NC(=O)[C@H](CCCNC(=N)N)NC(=O)[C@H](CCCNC(=N)N)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CCCNC(=N)N)NC(=O)[C@H](CCCNC(=N)N)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CCCNC(=N)N)NC(=O)CNC(=O)[C@H](CCCNC(=N)N)NC(=O)[C@H](CCCCN)NC(=O)[C@@H]1CCCN1C(=O)Cc1ccc(O)cc1)C(=O)O
InChI Key InChIKey=FPTNOAVHYADGRJ-UHFFFAOYSA-N
Data 1 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 1 hit for monomerid = 66879
Found 1 hit for monomerid = 66879
Affinity DataIC50: 230nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS...More data for this Ligand-Target Pair