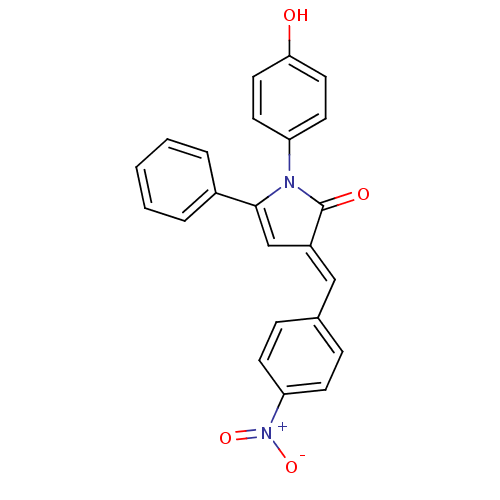
BDBM55412 (3E)-1-(4-hydroxyphenyl)-3-(4-nitrobenzylidene)-5-phenyl-2-pyrrolin-2-one::(3E)-1-(4-hydroxyphenyl)-3-[(4-nitrophenyl)methylidene]-5-phenyl-2-pyrrolone::(3E)-1-(4-hydroxyphenyl)-3-[(4-nitrophenyl)methylidene]-5-phenyl-pyrrol-2-one::(3E)-1-(4-hydroxyphenyl)-3-[(4-nitrophenyl)methylidene]-5-phenylpyrrol-2-one::MLS000521382::SMR000131790::cid_5346268
SMILES Oc1ccc(cc1)N1C(=O)\C(=C\c2ccc(cc2)[N+]([O-])=O)C=C1c1ccccc1
InChI Key InChIKey=GQDXXWHROQBAHL-UHFFFAOYSA-N
Data 3 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 55412
Found 3 hits for monomerid = 55412
Affinity DataIC50: 5.00E+4nMAssay Description:NIH Molecular Libraries Screening Centers Network [MLSCN] Emory Chemical Biology Discovery Center in MLSCN Assay provider: Susan Smith, Emory Univers...More data for this Ligand-Target Pair
TargetC-C chemokine receptor type 6(Human)
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 6.66E+4nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute(SBMRI, San Diego, CA...More data for this Ligand-Target Pair
TargetBeta-galactosidase(Escherichia coli)
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 6.66E+4nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute(SBMRI, San Diego, C...More data for this Ligand-Target Pair