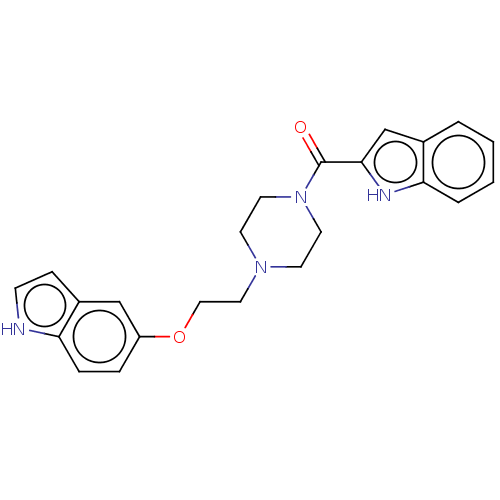
BDBM50528057 CHEMBL4550539::US11634404, Compound 16
SMILES O=C(N1CCN(CCOc2ccc3[nH]ccc3c2)CC1)c1cc2ccccc2[nH]1
InChI Key InChIKey=NYYVPFBZAVQNMH-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 50528057
Found 4 hits for monomerid = 50528057
Affinity DataEC50: 60.9nMAssay Description:Compounds were tested for their ability to compete with the orthosteric radioligand [3H]methylspiperone for binding to the D3 DAR using stable HEK ce...More data for this Ligand-Target Pair
TargetD(3) dopamine receptor(Human)
National Institute of Neurological Disorders and Stroke
Curated by ChEMBL
National Institute of Neurological Disorders and Stroke
Curated by ChEMBL
Affinity DataEC50: 61nMAssay Description:Agonist activity at human D3R expressed in CHOK1 cells assessed as induction of beta-arrestin recruitment measured after 90 mins by beta-galactosidas...More data for this Ligand-Target Pair
TargetD(2) dopamine receptor(Human)
National Institute of Neurological Disorders and Stroke
Curated by ChEMBL
National Institute of Neurological Disorders and Stroke
Curated by ChEMBL
Affinity DataIC50: 440nMAssay Description:Antagonist activity at human D2R expressed in CHOK1 cells assessed as inhibition of dopamine-induced beta-arrestin recruitment measured after 120 min...More data for this Ligand-Target Pair
TargetD(2) dopamine receptor(Human)
National Institute of Neurological Disorders and Stroke
Curated by ChEMBL
National Institute of Neurological Disorders and Stroke
Curated by ChEMBL
Affinity DataIC50: 440nMAssay Description:For a secondary-screen and selectivity assays, DAR PathHunter βarrestin GPCR cell lines from DiscoverX (Fremont, Calif.) were used. In the D2 Re...More data for this Ligand-Target Pair