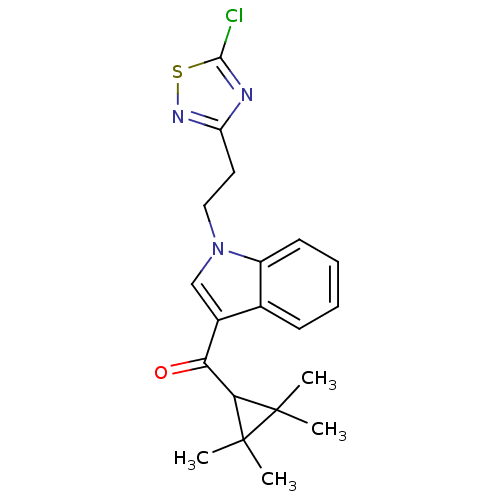
BDBM50303550 (1-(2-(5-Chloro-1,2,4-thiadiazol-3-yl)ethyl)-1H-indol-3-yl)(2,2,3,-3-tetramethylcyclopropyl)methanone::CHEMBL568796
SMILES CC1(C)C(C(=O)c2cn(CCc3nsc(Cl)n3)c3ccccc23)C1(C)C
InChI Key InChIKey=MQXCAMSGAGAYKD-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50303550
Found 3 hits for monomerid = 50303550
Affinity DataKi: 8.30nMAssay Description:Displacement of [3H]CP-55940 from human recombinant CB2 receptor expressed in HEK293 cellsMore data for this Ligand-Target Pair
Affinity DataEC50: 109nMAssay Description:Agonist activity at human recombinant CB2 receptor expressed in HEK293 cells coexpressing Galphaq/o5 assessed as calcium mobilization by FLIPR assayMore data for this Ligand-Target Pair
Affinity DataKi: 4.20E+3nMAssay Description:Displacement of [3H]CP-55940 from human recombinant CB1 receptor expressed in CHO cellsMore data for this Ligand-Target Pair